Health & Medicine
Newborn vaccine set to save lives

Researchers discover that a type of immune cell can cannibalise the properties of other cells, creating the potential for harnessing them for new therapies and vaccines
Published 22 February 2022
Melbourne researchers have discovered that an immune cell thought largely to be focused on fighting infection in newborns and infants, can in fact cannibalise the features of other immune cells, making it a potential new target for developing therapies across a range of infections.
Just published in Science, and a culmination of 15 years of work, the research by the Peter Doherty Institute for Infection and Immunity and the Bio21 Institute has found that so-called Marginal Zone (MZ) B cells can actually steal features from dendritic cells – the cells that act as sentinels alerting our immune system to infection.

If further research can uncover exactly how this happens then there is the potential for MZ B cells to be harnessed as a way to prompt more effective immune responses to specific infections.
“This finding is important because if we know what MZ B cells are really capable of doing, then we might be able to design vaccines or therapies to recruit them and boost our immune defences,” says University of Melbourne Professor Jose Villadangos, a laboratory head at the Doherty Institute and the Bio21 Institute and co-lead researcher on the paper.

Health & Medicine
Newborn vaccine set to save lives
MZ B cells, which produce antibodies that can attack a broad range of infections, and dendritic cells that orchestrate the immune response, both play highly important roles in the immune system, but until now it was believed they acted independently of one another.
“MZ B cells play a crucial role in protection against infection early in life, producing antibodies that can neutralise many types of bacteria in newborns and infants,” explains Professor Villadangos.
“As we develop, other cells take over this function and become the most prominent contributors to long-lasting immunity, but we believe the MZ B cells retain unique capabilities that may be useful in the fight against certain pathogens at any time during life.”
University of Melbourne Associate Professor Justine Mintern, a laboratory head at the Bio21 Institute and a co-author of the study, says the MZ B cell’s contribution to immune defence was thought to be limited because they appeared unable to perform some critical functions.

“In particular, we thought they were unable to coordinate the actions of different immune cell types, an ability we considered only dendritic cells to have,” says Associate Professor Mintern.
“Our research is contributing to change this perception, opening up new possibilities to harness the full potential of MZ B cells.”
Health & Medicine
Q&A: How immune cells could help diabetes and stroke
In their study, the researchers uncovered a new mechanism that enables MZ B cells to interact closely with dendritic cells and acquire the membrane proteins that allow dendritic cells to communicate with the foot soldiers of the immune system, T-cells, to trigger an immune response.
These membrane proteins, known as MHC molecules, are a highly specialised type of receptor that is exposed on the surface of dendritic cells.
The researchers discovered that MZ B cells interact with dendritic cells through the innate immune system. This is the immune system we are all born with and which can attack a broad range of infections, but unlike our adaptive immune system, it can’t change to attack other specific infections.
They found that a protein of the innate immune system called Complement C3 can bind with dendritic cells via the MHC receptors, and MZ B cells can then use these proteins to connect to dendritic cells.
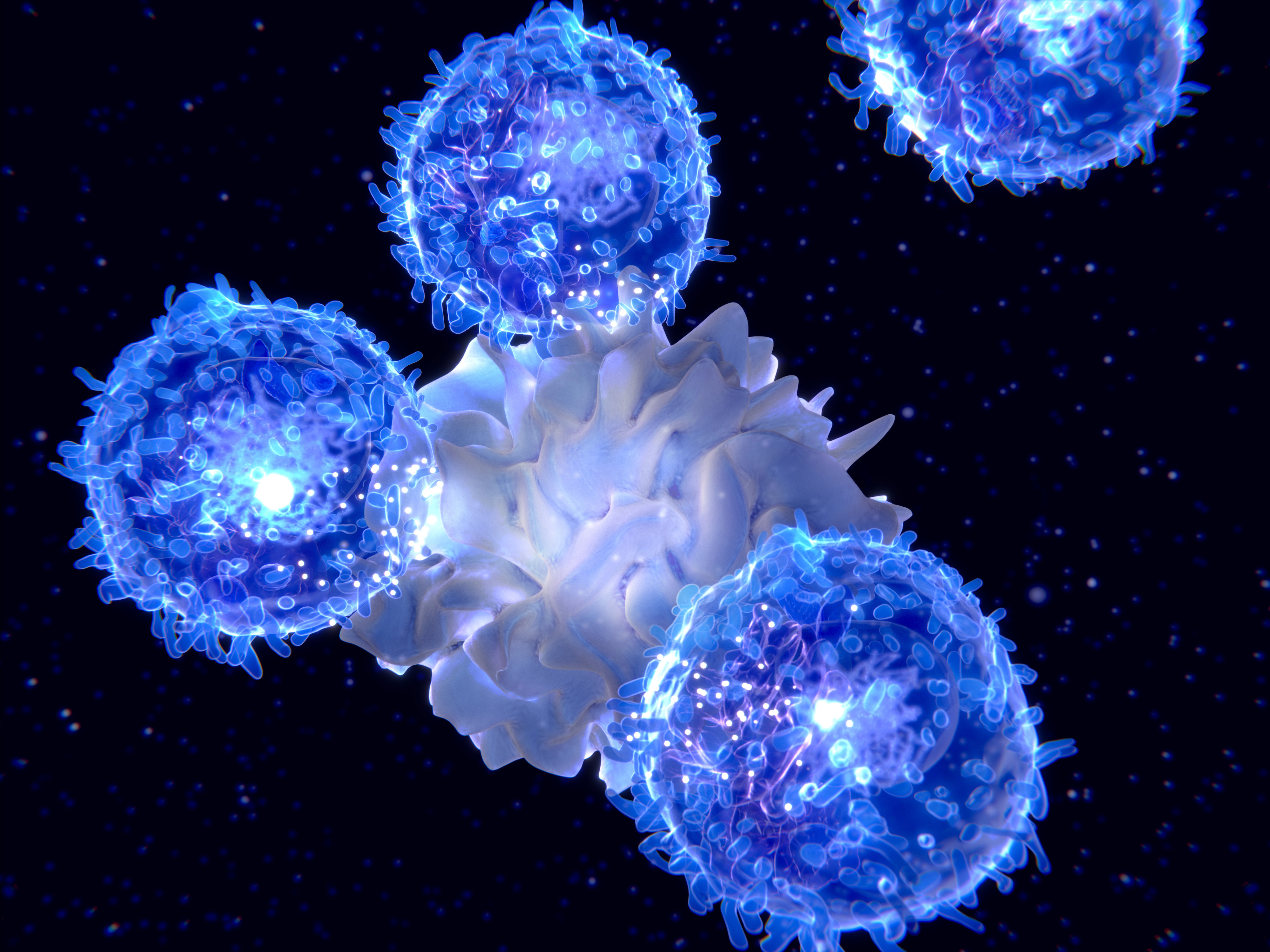
“We are the first to describe this association,” says Associate Professor Mintern.
Dr Patrick Schriek, lead author on the paper who was a PhD student in Professor Villadangos’ and Associate Professor Mintern’s laboratories at the time, not only performed the key experiments that led to this discovery, but was also responsible for the next crucial finding.
“Once Complement C3 has bound to the MHC receptors, MZ B cells use their own receptor to specifically bind to C3 and draw the dendritic cell close,” explains Dr Schriek.
Health & Medicine
Premature birth linked to lung disease later in life
“Then, something remarkable happens: the MZ B cell rips the surface of the dendritic cell – their plasma membrane – and incorporates it into their own. It is a form of cellular cannibalism known as trogocytosis.
“The outcome is that the MZ B cell can now use the MHC molecules captured from a dendritic cell to communicate with T cells. Effectively, the MZ B cell can now pretend to be a dendritic cell”.
The discovery has a surprising link to Laureate Professor Peter Doherty himself, Patron and namesake of the Doherty Institute.
“This finding was unexpected and particularly satisfying for us because Peter Doherty was awarded the Nobel Prize in 1996 for first describing the function of MHC molecules,” says Professor Villadangos.

“I have studied MHC molecules for over 30 years and I never expected we would stumble upon a new function carried out by these molecules, let alone that it would happen in an Institute named after Professor Doherty.”
Professor Villadangos says “The next challenge is to figure out how to recruit MZ B cells, exploit their newly-found properties and harness their largely unexplored capabilities.”

Health & Medicine
Cutting through the hype on platelet-rich plasma
“Our laboratories are engaged in multiple collaborations with academic and industry partners, testing and improving new vaccine designs and immunotherapies.”
“Ultimately the goal is to be able to use MZ B cells in a new way to fight infection,” says Associate Professor Mintern.
“We may be able to develop more effective strategies to prevent or fight infections using vaccines that don’t follow traditional rules but recruit cannibalistic MZ B cells to deliver new and improved outcomes.”
Banner: Getty Images