Health & Medicine
The tech boosting world-class antimicrobial research
Antimicrobial resistance is part of the chemical warfare bacteria wage in nature, and that’s where researchers now looking for new clues to overcome it
Published 29 March 2022
The growing resistance of bacteria to many of the antibiotic drugs we take for granted is now an urgent problem worldwide. Recently published research in the general medical journal, The Lancet, suggests that more than 1.2 million people died in 2019 because of antimicrobial resistance.
If antibiotic resistance is allowed to grow unabated, the number of deaths per year would balloon to an estimated 10 million by 2050. This will be considerably more than the 8.2 million people who currently die of cancer each year.

In response, medical researchers – including myself – have been racing to identify new antibiotics or modify existing antibiotics that are too toxic to use.
For example, in research recently published in Nature Communications my co-researchers and I have found a way to disconnect the toxicity from an existing but dangerous antibiotic in a step toward potentially making it usable.
But what hasn’t had enough attention is looking for answers from the microorganisms that make naturally occurring antibiotics.
While the scale of antimicrobial resistance may be new, the emergence of resistance isn’t at all new – in fact, it’s primordial.

Health & Medicine
The tech boosting world-class antimicrobial research
The majority of antibiotic medicines in clinical use today are derived from natural antibiotics that are produced by soil microbes to attack rival microbes in the microscopic war over access to nutrients – the most famous example is penicillin.
So antibiotics are ancient, dating back hundreds of millions of years, and resistance is equally ancient, reflecting the continuous co-evolution of small molecules in natural environments and microbial genomes.
For example multi-drug resistant bacteria were discovered growing on the walls of the Lechuguilla Cave in the US, a habitat that has been isolated from the surface for more than four million years.
Our contemporary line of thought is that the rise and spread of resistance is a direct result of the clinical use and/or misuse of antibiotics that continuously challenges bacteria to evolve resistance. However, it’s becoming increasing apparent that resistance is a natural phenomenon that simply re-emerges in a clinical setting.
Fundamentally then our contemporary understanding of the mechanisms of antimicrobial resistance is being taken ‘out of context’.
We have been focused on looking at the emergence of resistance in clinical settings, but we have virtually no understanding of how bacteria develop resistance to, and respond to, antibiotics in the ‘wild’ where resistance has been evolving over millennia as a result of microbe-on-microbe chemical warfare.

For example, polymyxins are naturally occurring antibiotics that are used to treat difficult infections like sepsis and central nervous system brain infections. Polymyxins are effective against Gram-negative bacteria which have a tough outer membrane barrier – the result of bacteria constantly evolving to protect themselves from the noxious chemicals they may encounter in their environment, like antibiotics.
The success of polymyxins and, conversely, the critical risk to human health of bacteria evolving resistance to polymyxins, has meant that research in this area has primarily focused on their role in clinical settings.
In contrast, the function of these antibiotics in natural environments has received almost no attention. It means we are woefully ignorant of the role of antimicrobial resistance in nature.
Health & Medicine
A new weapon in the war against superbugs
Over 80 per cent of antibiotics in clinical use are naturally occurring chemicals that are made by soil bacteria. In the wild, polymyxins are molecules produced by the soil microbe Paenibacillus polymyxa to kill Gram-negative bacteria that invade its environment in competition for nutrients.
If we can better understand the complex picture of how antibiotic resistance evolves in nature, it will help make clearer the processes that lead to bacterial pathogens developing resistance to polymyxins – and so help us find ways to control it.
This is where the study of environmental microorganisms that produce antibiotics, like Paenibacillus polymyxa, and their interactions with pathogenic Gram-negative bacteria, is critical.
My team is investigating how and why bacteria develop resistance to polymyxins under conditions that mimic nature. Importantly, this will allow us to compare what happens in nature with how pathogenic bacteria develop resistance in hospitals.
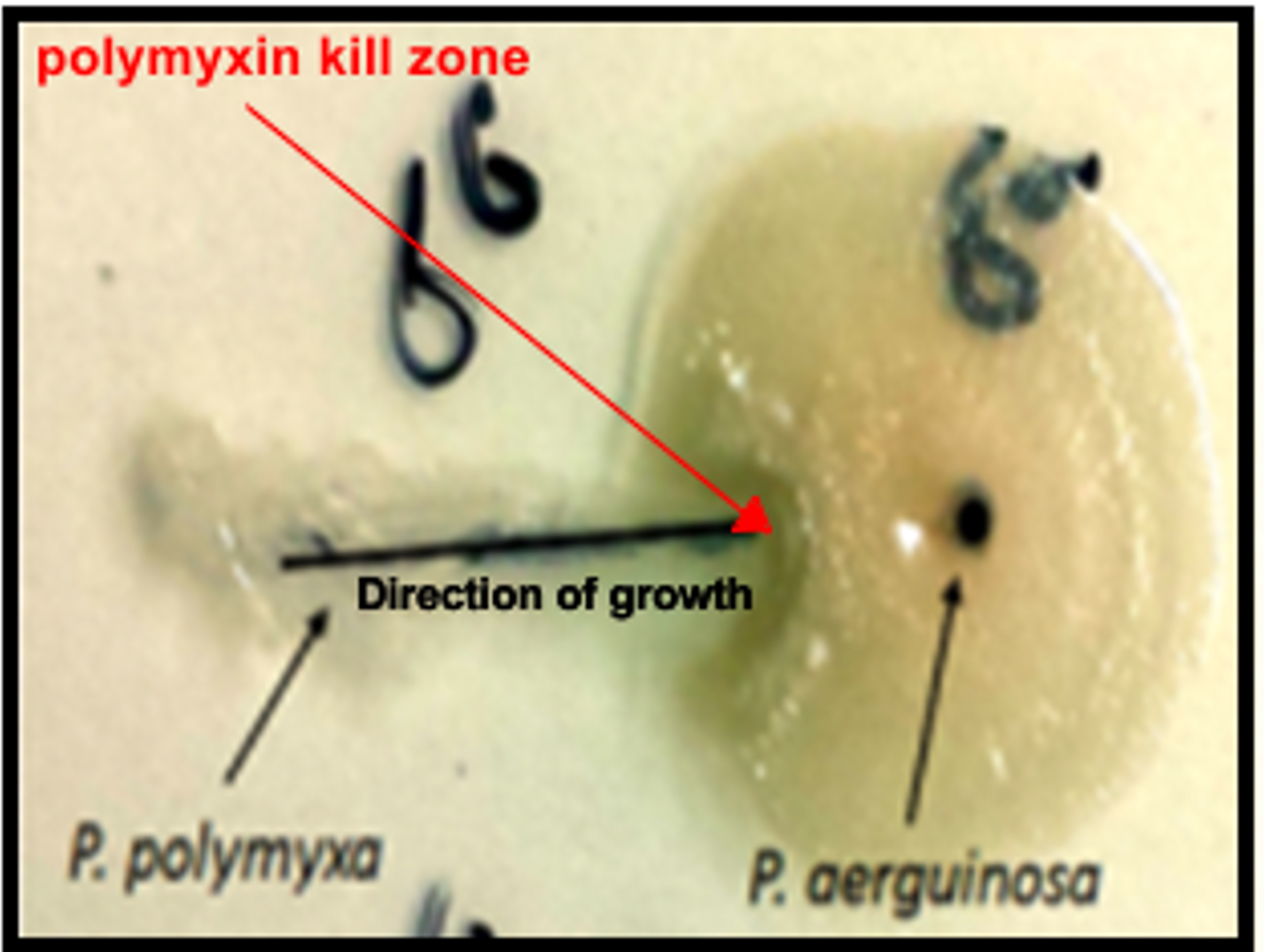
At the moment there is a major disconnect in our understanding of how antibiotic resistance mechanisms that evolve in the clinical setting relate to mechanisms of resistance that have emerged in nature over countless millennia.
Antibiotic producing soil microbes not only represent sources of antibiotics but also reservoirs of resistance genes, such that changes in these species might be relevant for the passage of previously unknown resistance determinants into human pathogens.
Health & Medicine
The chemical warfare against bacterial superbugs
We aim to provide the first understanding of polymyxin resistance in natural environments, which may eventually help us to better predict and counteract the emergence and future evolution of the antibiotic resistance that is plaguing our community and hospitals.
In January, Associate Professor Tony Velkov was awarded a REDI (Researcher Exchange and Development with Industry) Fellowship from independent Federal Government agency MTPConnect. Under the fellowship he will undertake a 12-month project with US-based Qpex Biopharma towards developing new polymyxin antibiotics to combat increasing antibiotic resistance. The data were recently published in Nature Communications journal.
His research programs are funded by his three five-year grants for US National Institute of Health (2016, 2019 and 2022) amounting to around $US7.8 million, a grant from the US Department of Defense of around $US1 million, and a five year Project Grant from the National Health and Medical Research Council amounting to around $A900,000.
Banner: Getty Images