
Health & Medicine
Using chemotherapy to fight malaria

An international collaboration has developed a new drug candidate that stops the malaria parasite from breaking down waste, resulting in fatal ‘molecular constipation’ and hope for new treatments
Published 20 October 2021
Diseases caused by infectious organisms pose an enormous threat to global health, food security and sustainable development, and malaria is one of the worst of these diseases, causing more than 400,000 deaths each year.
Sadly, delays related to the COVID-19 pandemic are expected to impede efforts to treat and prevent malaria. Even more alarmingly, current antimalarial treatments are rapidly losing their effectiveness as the different malaria parasites develop resistance.

A malaria vaccine has been approved recently but shows only about 30 per cent effectiveness against severe disease and urgently needs to be augmented with new treatments with new modes of action.

Health & Medicine
Using chemotherapy to fight malaria
Professor Leann Tilley and colleagues from the Department of Biochemistry and Pharmacology at the University of Melbourne, have co-led a large international effort to develop a new type of drug that is specific for the malaria parasite Plasmodium falciparum, which along with the Plasmodium vivax malaria parasite poses the greatest threat to humans.
Working with a large pharmaceutical company, Takeda Pharmaceuticals, the team started investigating compounds that Takeda had developed to treat cancer.
These inhibitor compounds target a sophisticated enzyme complex, called the proteasome, that both human cells and plasmodium rely on to break down unneeded or damaged proteins.
They are particularly effective in cancer therapy because cancers cells grow at a gangbuster rate, generating lots of damaged proteins, so blocking this process leads to cancer cell death.
The team designed more than 50 new versions of the inhibitors by tweaking the chemical structure, with the aim of finding compounds that were potent against Plasmodium falciparum but no longer toxic to human cells.
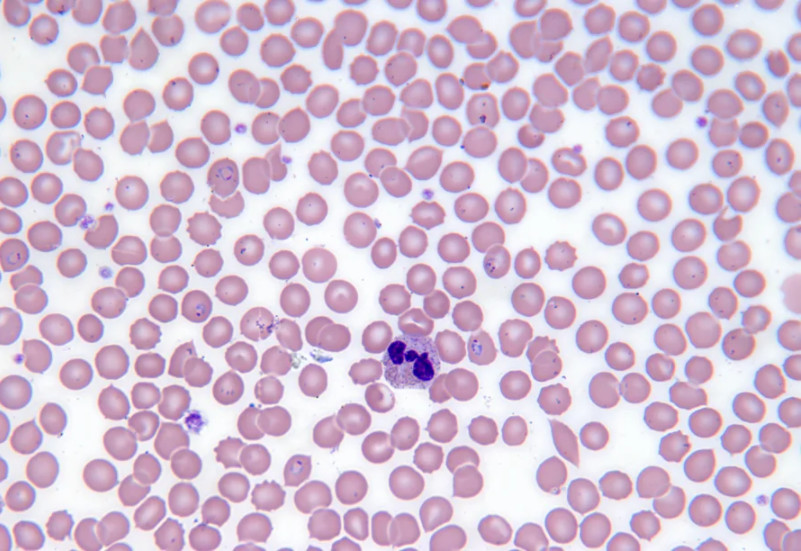
After four years of work, involving dozens of researchers based across five continents, the team found a compound that is potent, selective, fast-acting, and active at all stages of Plasmodium falciparum’s lifecycle (both inside the mosquito and the human host).

Sciences & Technology
The genes that turn malaria into a killer
“We were very excited”, says Professor Tilley. “Most importantly, our work showed that the compound could cure malaria in mouse models infected with human malaria parasites.”
Next, Dr Stanley Xie, also from the University of Melbourne, undertook some biochemical detective work to confirm that the compound works by inhibiting the plasmodium proteasome; but was now much less active against the human proteasome.
He showed that inhibition of the parasite proteasome prevents the parasite from degrading its excess proteins. This results in a form of molecular constipation, clogging the parasite’s recycling system and ultimately, leading to cell death.
But the team still didn’t understand why the new compound was so effective at specifically targeting the malaria parasite proteasome. So the next step was to analyse the new compound using the Bio21 Institute’s cryo-electron microscope (cryoEM).
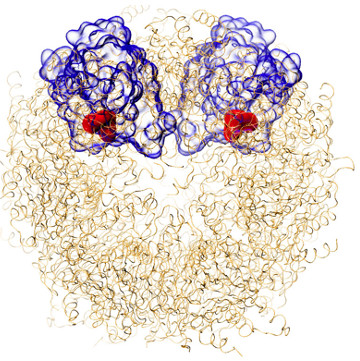
This new technique capitalises on a recent revolution in structural biology where samples, including proteins and viruses, can be flash-frozen and bombarded with electrons to produce microscopic images of their molecular structure.
Health & Medicine
Exposing malaria’s atomic machinery
Professor Eric Hanssen captured thousands of images of proteasome particles bound to the drug, suspended in amorphous ice, at 50,000-times magnification.
Then, colleagues Dr Riley Metcalfe, Dr Craig Morton and Associate Professor Mike Griffin undertook the daunting task of fitting and refining approximately 8,000 amino acids in the complex to generate the 3D structure.
What emerged was a detailed molecular picture of the “poisoned” proteasome. “It was amazing to finally look the inhibited proteasome in the face”, says Professor Tilley.
Associate Professor Griffin adds: “We could see that the new inhibitor binds to the proteasome in a region of the enzyme’s active site that is structurally different in the human proteasome. This molecular nuance underpins the selective binding of the new inhibitor only to the malarial target”.
A particularly rewarding part of the work, says Professor Tilley, was the ability to work with a large and highly collaborative team, bringing together scientists from academia, the pharmaceutical industry and the not-for-profit malaria drug development peak body, Medicines for Malaria Venture.

Health & Medicine
Immunity to COVID-19: Lessons from malaria
“The work will help develop new antimalarial therapeutics. The next step is to work on improving the lifetime of the drug in the bloodstream so that the treatment can be used to cure malaria in humans with a single tablet.
The next phase of the work will include the Bio21 Institute’s new $A20 million Titan Krios cryo-electron microscope, housed in the Ian Holmes Imaging Centre. This cutting edge microscope was co-funded with precinct partners.
“The improved resolution and higher throughput offered by the Krios will greatly facilitate the drug discovery pipeline,” says Bio21 Director, Professor Michael Parker.
“We are excited about the possibilities and hopeful that the work will also underpin the development of new inhibitors for other infectious diseases using similar ideas,” Professor Tilley says.
Banner: Getty Images