
Health & Medicine
Our genetic strength in numbers

Using stem cell modelling, researchers have developed genetic roadmaps for two of the world’s leading causes of irreversible blindness
Published 31 July 2022
Distorted or blurred sight, black or dark spots and potential loss of vision – these are symptoms of primary open-angle glaucoma (POAG) and age-related macular degeneration (AMD), which are predicted to globally affect more than 110 million people and 288 million people respectively by 2040.
There are no cures and no current definitive treatments for both POAG and for the dry form of AMD – it is simply a matter of trying to preserve people’s vision for as long as possible.
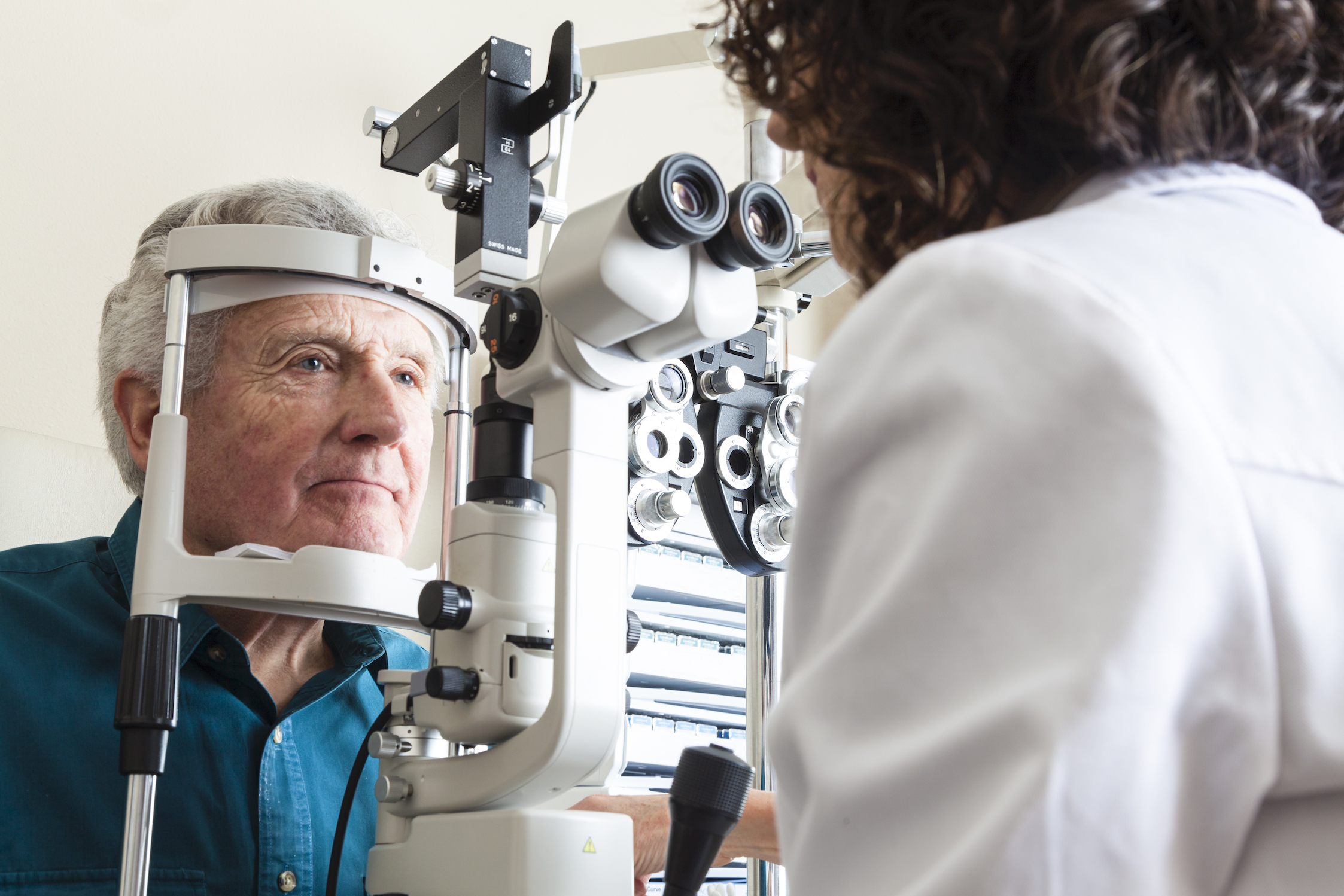
The exact pathogenic mechanisms – that is, the disease pathways – aren’t well understood, despite intense international research efforts. This has been largely due to our lack of suitable experimental models, making the task of identifying and testing treatments significantly more difficult.
Having access to pathologically-relevant models of human optic nerve or retinal degeneration is crucial for understanding, screening and developing accurate treatment strategies that prevent or slow progression of these blinding diseases.

Health & Medicine
Our genetic strength in numbers
Human induced pluripotent stem cells (iPSCs) are a powerful tool to investigate these types of conditions.
These stem cells, which we produce by reprogramming somatic cells like skin or blood cells from selected individuals or specific genetic background, can be grown into any specific cell type of interest, to make “a biopsy in a dish” and generate an effective disease model for therapy development.
This is particularly suited to the modelling of optic nerve and retinal conditions, where using patient skin can be made into stem cells and then guided to become the cells affected in glaucoma or AMD. This technique overcomes the difficulties in accessing these tissues in living individuals.
Together with Professor Alex Hewitt from the Centre for Eye Research Australia and University of Tasmania, and Professor Joseph Powell’s teams at the Garvan Institute of Medical Research, we took this approach in the hope of contributing to the discovery of treatments using a disease model based around patients’ own stem cells.

In our latest research, we have bridged a gap in the knowledge around one form of glaucoma, called Primary Open Angle Glaucoma (POAG), which affects the retinal ganglion cells that form the optic nerve; and the end stage of AMD called geographic atrophy in which retinal pigment epithelium cells die.
Our work – one of the largest and most detailed stem cell modelling studies for any disease – was published in Cell Genomics.

Health & Medicine
Taking on blood cancer - actively
By comparing stem cell models of the retinal ganglion cells of people with POAG to those without the disease, 312 novel genetic features of these cells were uncovered. These unique genetic features provide hundreds of new targets for researchers developing drugs to treat glaucoma.
Our second study, published in Nature Communications, also uses patients’ own stem cells, and uncovered novel genes and pathways most likely involved in AMD – including in mitochondrial activity and energy production.
The findings are the result of a decade-long national collaboration led by Professor Hewitt, Professor Powell and his Garvan Institute colleague Anne Senabouth, and our team at University of Melbourne, including Dr Maciej Daniszewski and Dr Grace Lidgerwood.
The research relies on reprogramming, which is a special way to make stem cells that is based on the work of 2012 Nobel Prize winner Professor Shinya Yamanaka.

His team demonstrated that one could take any “somatic” cell of our body, like skin cells, and reprogram them into becoming stem cells – or “pluripotent” – again.
These pluripotent cells are called iPSCs. Once obtained, they can theoretically become any cell type of the body, including cells of the eye that are affected in POAG and AMD.
These are the first demonstrations of how large banks of iPSCs can be used to model complex diseases and could pave the way for the study and discovery of other conditions lacking reliable models and treatments – like forms of dementia or diabetes.

Health & Medicine
Living with a rare genetic disorder
Human iPSC models have been shown to be very useful tools to understand monogenic diseases, including developmental diseases, where mutations in one gene drive the disease.
However, their use for the modelling of complex diseases, where multiple genetic factors and environmental influences, like ageing, might play roles was unclear and controversial.
This is partially because stem cell derivatives remain “young” in the dish, so it had been argued that these cells cannot model diseases associated with ageing.
We were curious to see if we could, indeed, use these cells to model complex diseases that affect so many of us, including POAG and AMD. We hypothesised that cell derivatives obtained from each patient stem cells – retinal ganglion cells for glaucoma and retinal pigment epithelium for AMD – would show specific “disease signatures” when compared to healthy derivatives.
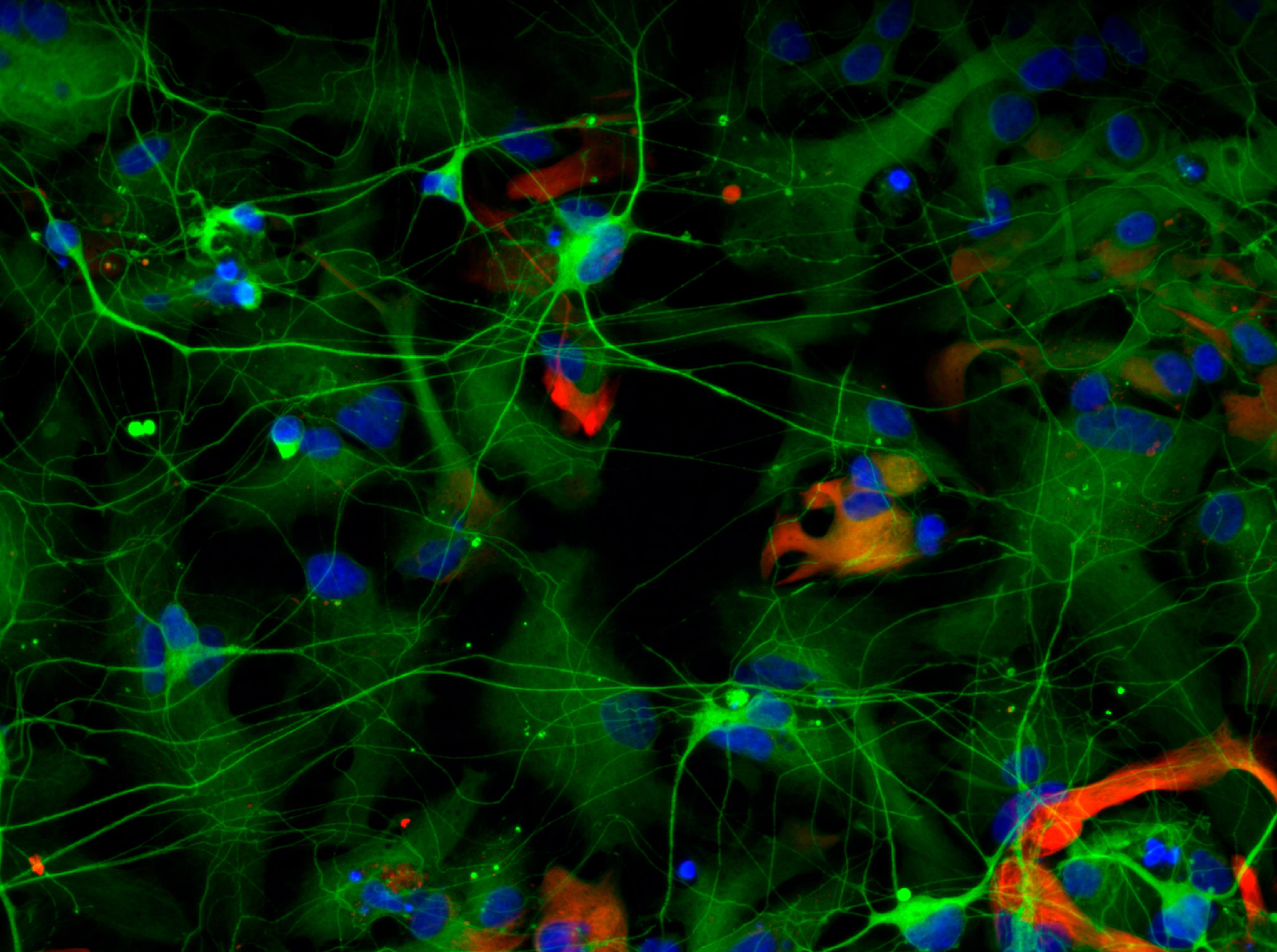
Over the past 10 years, we relentlessly collected skin biopsies kindly contributed to our research by hundreds of individuals – with POAG or AMD and healthy controls.
We generated iPSCs from them, established reliable protocols of differentiation of these cells into our cells of interest, and optimised powerful and reliable methods of assessments of molecular signatures.
We believed those could be observed by transcriptomics analysis, which is when we precisely look at the gene regulation in each sample, using single cell RNA sequencing and followed by complex computational biology analysis.
Health & Medicine
Uncovering the triggers of severe COVID-19 in kids
The combination of approaches is the novelty of our work – making patient stem cells in large numbers, using them to model diseases that come late in life, and then taking a transcriptomic approach as a readout.
Now that we have demonstrated that the approach works in models of glaucoma and AMD, we can start looking for potential targets that contribute to the discovery of treatments for these degenerative diseases.
Specifically, we want to continue improving our understanding of the genetics of these neurodegenerative diseases, with the ultimate hope that this research will facilitate preclinical trials and translatable outcomes for so many people in our community.
This research was the result of an extensive national collaboration, including researchers from the University of Melbourne, Garvan Institute of Medical Research, Centre for Eye Research Australia, University of Tasmania, QIMR Berghofer Medical Research Institute, St Vincent’s Institute of Medical Research, Flinders University, Lion’s Eye Institute, Macquarie University, Menzies Institute for Medical Research, and University of New South Wales.
National Health and Medical Research Council, Australian Research Council, Macular Disease Foundation Australia, the Ophthalmic Research Institute of Australia, the Joan and Peter Clemenger Foundation, the Bright Focus Foundation, Philip Neal bequest, Stem Cells Australia and the University of Melbourne.
Banner: Fluorescent imaging of retinal pigment epithelium (RPE) cells showing each cell’s nucleus (blue), and markers of RPE cells (purple and Yellow). Picture: Jenna Hall/University of Melbourne