
Health & Medicine
Why are women ignored by medical research?
Mechanobiology brings together engineering, biology, and physics – and this technology could help predict your risk of developing a musculoskeletal condition
Published 7 March 2020
Musculoskeletal conditions like arthritis and osteoporosis are considered the fourth most significant disease burden in Australia.
These chronic conditions are often painful and disabling for sufferers – flare ups are uncomfortable and have a large impact on quality of life, including the ability to do simple chores and a person’s capacity to work.

This group of musculoskeletal conditions represents around a $A55.1 billion annual cost to the Australian economy, including direct health costs, lost productivity and reduced quality of life; but has a low direct mortality rate.
This is a significant and affected population which only receives a small percentage of Australian research funding.
This is a problem, particularly with a predicted 43 per cent growth in musculoskeletal cases projected over the next two decades, including a surge in the number of older Australians living with the conditions.

Health & Medicine
Why are women ignored by medical research?
My research, as leader of the Integrative Cartilage Research Group in the Department of Biomedical Engineering, is working to develop and apply innovative technologies to explore and maintain our musculoskeletal health.
We are developing a range of techniques that help interpret how joints respond to stresses that ultimately could allow us to predict someone’s risk of developing a musculoskeletal condition.
Mechanobiology is a multidisciplinary field that encompasses cell and developmental biology, bioengineering and biophysics.
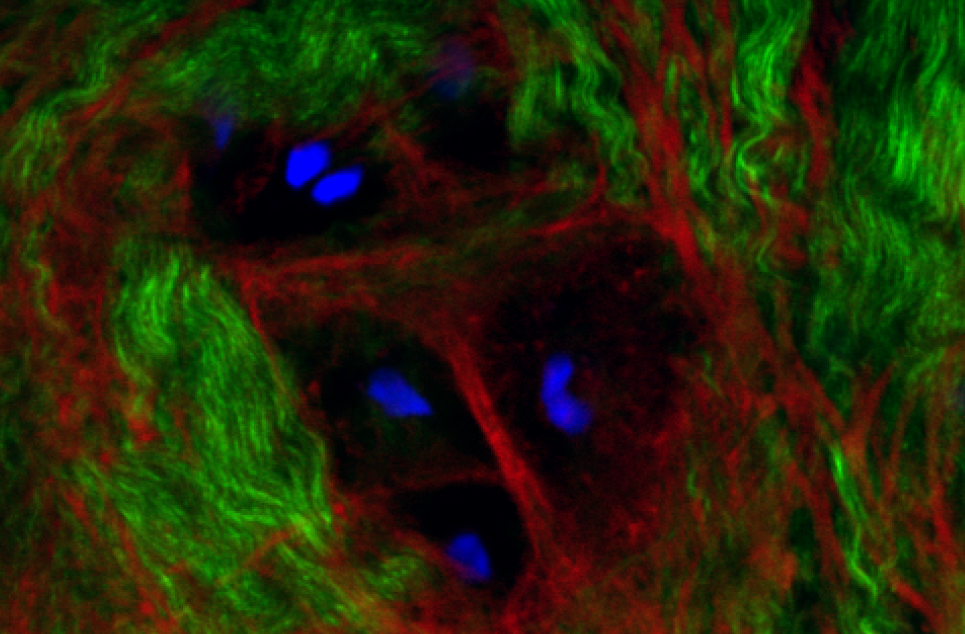
It focuses on the dynamic interaction between cells and their environment; specifically, from mechanical forces.
When you walk your body has complex systems working in an almost choreographed dance to ensure you have a smooth, comfortable motion. This includes specifically designed parts all working together – including weight-bearing muscles, ligaments, bones, cartilage, interjoint fluids and internal support muscles.
My lab focuses on how these individual parts are designed, respond to and work together to deal with different activities.
How our cells and tissues are structured and formed is related to their job in your body.
Health & Medicine
Fighting infection with curiosity
Your ear is cartilage, but it is designed at a molecular level to do a very different task than, say, cartilage in your knees. The architecture of cartilage is very important to how it works as part of the system in specific parts of your body
Our aim is to reveal how cells in the cartilage respond to these forces, like movement or gravity, and the influence of the tissue architecture on that response.
We call this ‘mechanobiological signalling’ and we aim to use it to investigate cartilage and joint disorders.
Using engineering and software design, imaging physics and musculoskeletal biology, we investigate these complex cartilage and joint systems from the smallest element (the cells) through to the whole organ (multiple types of cells working together).
This approach is called multiscale mechanobiology.
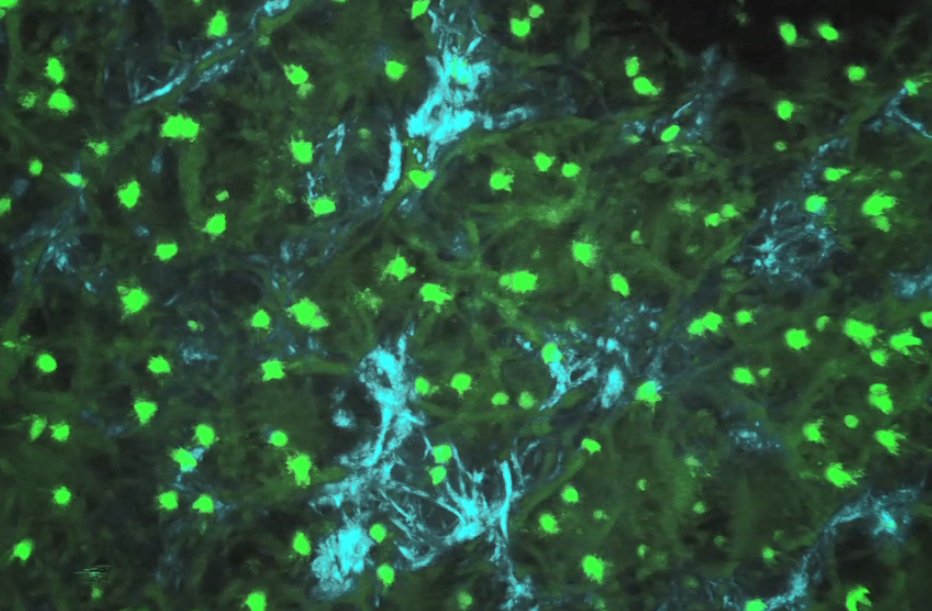
Our research develops new approaches that captures a suite of measures and markers for musculoskeletal conditions in one platform. We can measure specific incidents, how tissues change structure and relationship with organs over time, or how tissues, cells and organs respond all at once.
Essentially, it’s a grand engineering challenge.
The goal is to provide innovative technologies for diagnosis, treatment and prevention of cartilage and musculoskeletal joint disorders.
This would allow people to slow down progression, better manage their conditions and associated symptoms, and potentially prevent onset of some of these chronic debilitating conditions.

Our cells, tissues and organs are in constant movement – they’re communicating, moving, growing, repairing and dying while still supporting our heads and allowing us to climb stairs.
How are they able to function under these often-unpredictable stresses and movements, and continue to repair and grow?
To understand this, our team has developed a device called Felix which allows us to look at materials through a microscope while we also apply a stress.
Felix allows us to study cell-matrix remodelling (how cells group and support themselves to become a tissue or organ), providing an accurate measurement of how biological materials remodel and grow over time.

This gives us new targets for making materials that can be implanted into the body to replace those that don’t work anymore. If we can replicate how these cells organise and support themselves sustainably to form tissues, we can synthetically replicate this and implant them.
Another tool is micro-computed tomography (microCT), which is essentially a three-dimensional x-ray that allows us to study the whole living joint over time and see how the bone remodels with the onset and progression of arthritis.
MicroCT is a smaller version of the clinical CAT scanners you see in hospitals and is used to get very high-resolution images of bone structures.
We can use it to scan samples over time to get very detailed structural information of how the architecture of the structures is influenced by daily loads. With this information we can identify what loads are good (or bad) for healthy joints and how the accumulative stress affects them.

Arts & Culture
Motherhood in a climate crisis
We are also developing new contrast agents – substances used to increase the contrast of structures or fluids within the body in medical imaging – that add definition and details to better differentiate cartilage and bone in a 3D image.
Using time-lapse imaging, we can then investigate the homeostatic balance between these two tissues that are vital to maintaining joint integrity.
Cartilage and bone are very different – cartilage is soft and bone is hard – but they are connected. We all this ‘homeostatic balance’ which describes how cartilage and bone balance each other and use each other to stay healthy.
Given the multi-faceted characteristics of arthritis, it is critical to identify disease-specific features that disrupt this balance and could cause tissue damage and disease onset.

But despite decades of intensive research, the disease-specific features of arthritis initiation and development remain poorly understood.
Imaging is a very useful tool. But more valuable are the computational approaches that can be used to extract 3D quantitative numbers linked to the disease.
These numbers can tell us how much bone we have gained or lost as a disease takes hold, or whether the cartilage is wearing away in a specific region, or whether we have changed the way we walk because of these changes.

Business & Economics
Women, age discrimination and work
Our team have developed a first-of-its-kind quantitative morphometric analysis (QMA) software which can extract this data related to structural change that we can see from medical imaging like CT scans.
QMA is a powerful tool for understanding the development of the disease. It has the potential for performing accurate diagnosis and helping to develop appropriate treatments.
Musculoskeletal conditions like arthritis and osteoporosis can have huge impacts on the control and quality of patients’ lives.
By working to understand how cells and tissues work together to handle stresses, we can design new technologies like implantable materials. But importantly, we can work towards understanding the progression, severity and, hopefully one day, the prevention of musculoskeletal diseases.
Banner: Getty Images