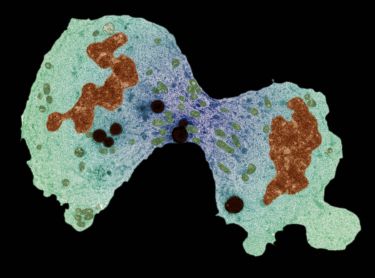
Health & Medicine
Our cancer preventing genes revealed
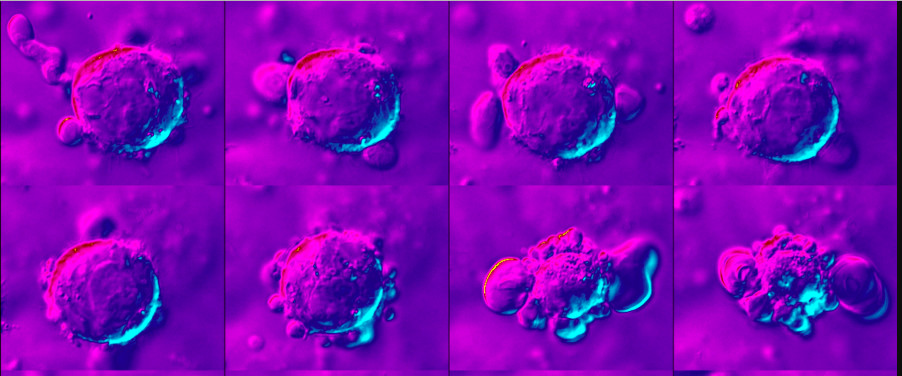
Australian scientists have developed a world-first compound that keeps cells alive and functioning when they otherwise would have died
Published 8 October 2019
The idea that our cells are set to ‘self-destruct’ is not entirely comforting.
But like all things, it is a question of balance.
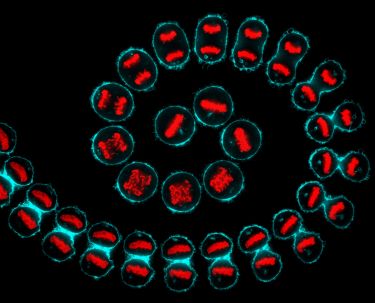
Known as apoptosis, this process of programmed cell death is essential for keeping the body healthy; it’s triggered when cells are aged and diseased as well as during development.
In the womb, our hands begin as mittens, so apoptosis directs cells between the fingers to die off, thereby forming our digits.
Fast-growing cells like our skin and gut-lining, which also faces strong digestive acids, are replaced most often. The average adult human loses between 50 and 70 billion cells each day due to apoptosis.

Health & Medicine
Our cancer preventing genes revealed
Because programmed cell death is irreversible once it has begun, apoptosis is tightly regulated by the body, and this regulation has been the focus of much research as its implicated in the progression of many diseases.
Excessive apoptosis can cause tissues to waste away or atrophy in conditions like muscular dystrophy, whereas an insufficient amount can lead to uncontrolled cell proliferation or growth, resulting in cancer.
Some proteins known as caspases get switched on to promote apoptosis, but this pivotal step is controlled by members of the BCL-2 protein family.
This family of proteins has been the focus of 11 years of collaborative research at the Walter and Eliza Hall Institute of Medical Research, led by Professor David Huang, Professor Guillaume Lessene and Professor Benjamin Kile (now at Monash University).
The team has recently published preclinical findings in the journal Nature Chemical Biology, highlighting their discovery of a unique compound that can keep cells alive and functioning when they otherwise would have died.
Within the BCL-2 family of proteins some promote cell survival, while others drive cell death.
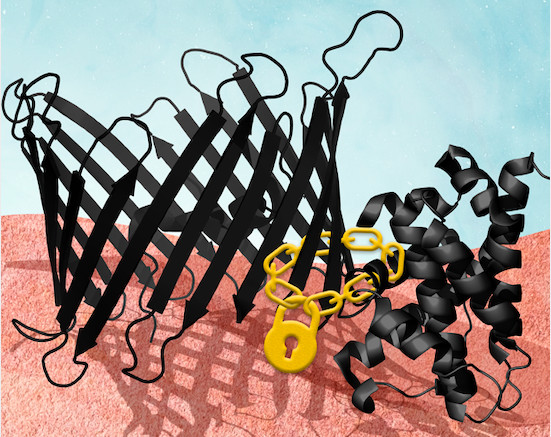
Proteins called BAK and BAX are involved in a critical step of cell death known as the ‘point of no return’ because cells are committed to die once either BAK or BAX is activated.
Health & Medicine
World-first test could predict your risk of a heart attack
So, key to the team’s research was understanding how to block BAK activation.
They screened an assembled collection of 250,000 potential small drug molecules for ones that blocked apoptosis.
“Our experiment pushed the cells right to the brink of death and looked for compounds that rescued it,” Professor Lessene explains.
“What we found was that when a compound we named WEHI-9625 binds to the protein VDAC2 in the apoptosis pathway, then BAK can no longer trigger cell death,” he says.
“It’s a bit like a when a person under arrest is handcuffed to a police officer, they are unable to run away or do anything much at all.”
The compound WEHI-9625 is now considered a proof-of-concept drug, and the next step for the team is to understand how it interacts with VDAC2 and BAK at a molecular level.
The finding could give clinicians the ability to intervene and prevent cell death, says Professor Lessene.
The new ‘cell death blocker’ is revolutionary in its ability to keep cells alive and healthy in the laboratory, he says.

Health & Medicine
Targeting ovarian cancer
“This would be game-changing for medical emergencies and procedures, such as minimising cellular damage after heart attacks, or preserving organs for transplants.”
Professor Kile, Head of Anatomy and Developmental Biology at the Monash Biomedicine Discovery Institute, says that WEHI-9625 is the first compound identified to successfully override apoptosis and keep cells functioning.”
“We have shown it is possible to halt the biochemical cascade that triggers cell death, right at the point where it begins”.
Professor Huang, a laboratory head in the Institute’s Blood Cells and Blood Cancers division, adds that the ability to stop unwanted cell death could be invaluable for the future of medical care.
“Acute injury can cause cells to die rapidly leading to the loss and weakening of tissues and muscles. In such circumstances, being able to prevent uncontrolled cell death could improve a patient’s recovery, or even their chances of survival,” he says.
“The Institute’s research into cell death spans more than 30 years, beginning with the landmark discovery in the late 1980s that the protein BCL-2 could enable prolonged cancer cell survival. This critical discovery helped to inform the development of an anti-cancer treatment for patients with leukaemia.”
“Our latest research shines light on ‘the other side of the same coin’, offering hope that one day drugs that successfully intervene to block apoptosis could be used to treat conditions such as cardiovascular diseases and degenerative disorders.”
Health & Medicine
Putting cancer cells to sleep
The researchers are now looking to apply the knowledge to developing cell death blockers that are effective and safe in humans.
Professor Huang says the next steps would also involve applying the knowledge to more advanced models of disease.
“There could be applications for keeping cells alive to prevent degenerative diseases,” he says.
The research was supported by the Australian Cancer Research Foundation, Australian National Health and Medical Research Council, Brownless Trust, DHB Foundation, HEARing CRC, MuriGen Therapeutics, Sylvia and Charles Viertel Foundation and the Victorian Government.
Banner Image: Human HeLa cancer cell, apoptosis. Credit: Paul Andrews/Univ. Dundee. CC BY. Wellcome Collection.