Health & Medicine
The Global Cancer Atlas

Mapping the human genome is revolutionising medicine. The aim? To turn cancer into a manageable, chronic disease like diabetes
Published 11 February 2020
Imagine ten jigsaw puzzles, each with 150 million pieces, jumbled together in a huge box.
The jigsaws are all double-sided, with a different picture on each side. The images on them are bland and one in 100 pieces has the wrong picture.

Despite all of this, you have just two days to complete the puzzles.
Tricky enough for you?
This is essentially what scientists face – figuratively speaking – each time they sequence a human genome. When they do solve the puzzle, just four of the pieces hold the key information they need.

Health & Medicine
The Global Cancer Atlas
It is an incredibly difficult and precise science, but mapping the human genome is revolutionising medicine, and researchers hope that it will help them to turn cancer into a manageable chronic disease like diabetes.
Until relatively recently, this sort of thing was a pipe dream.
The game changed significantly, however, when scientists announced that they had mapped the human genome in 2003. Since then, many others around the world have explored ways to use this ground-breaking information.
The potential for using an individual’s genetic information to personalise their medical treatment is enormous. But what exactly is the human genome and how do you sequence it?
The genome is an organism’s entire chemical blueprint. It is held as DNA in all cells with a nucleus and scientists are increasingly focusing on sequencing, or reading, genomes to understand the genetic functioning and causes of disease.

The human genome contains about three billion base chemical pairs, making it extremely difficult to read accurately. But that hasn’t stopped University of Melbourne researchers from using the technology to help cancer patients and those at risk of developing it.
The information is already being used to better understand why a person is at risk or to pinpoint how each patient should be treated. Well-targeted treatments increase the chances of success and reduce the chances of failure and/or debilitating side effects.
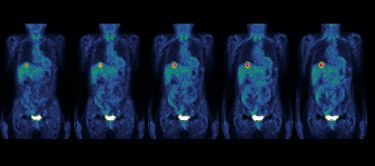
Professor Sean Grimmond, the Bertalli Chair in Cancer Medicine at the University of Melbourne’s Centre for Cancer Research (UMCCR), is focusing on rare and challenging cancers, such as pancreatic cancer.

Health & Medicine
Wiping cancer from our hard drives
People with pancreatic cancer have a five-year survival rate of less than 10 per cent.
The UMCCR is focusing on cancers of unmet need. These are the most challenging to treat cases, including rare or aggressive tumours, those resistant to standard therapies, or those that are traditionally difficult to diagnose.
When Professor Grimmond’s team sequenced the genome of its first Australian cancer patient nine years ago, it took more than six months and cost about $A1 million.
Using new NovaSeq 6000 machines and the latest computer technology, it now costs the same as an MRI scan.
The science is cutting edge, but if you’re picturing space-age shiny metals, rows of glowing buttons or laser lights then think again.

The Illumina NovaSeq machines that could save many lives resemble large, sleek photocopiers, with cartridges of reagents instead of ink to facilitate the sequencing process.
They live in the Victorian Comprehensive Cancer Centre, a building bringing together those at the forefront of world cancer research and treatment. Operated by the UMCCR, each machine has the capacity to sequence 50 human genomes in less than two days.
So, how do they do it?

Health & Medicine
Gene genies: Meet the researchers mapping our DNA to combat cancer
Sequencing a cancer patient’s genome essentially produces a genetic blueprint of their tumour. Initially, scientists take a tiny sample of the cancer, extract the DNA and process it before compressing it onto a slide.
The slide is fed into the sequencing machine, which produces terabytes of data that can only be decoded in pieces that are up to 150 bits long.
Scientists take those pieces and “smash them into random bits” to create a complex map that can be decoded. Hence, the jigsaw analogy.
“Before you start, you draw lines between the pieces and come back to use those clues,” Professor Grimmond explains. “It’s computationally very difficult. We can think of those bits as a computer hard drive – your DNA is like a three-gig hard drive.”
As well as mapping individual genomes, Professor Grimmond wants to build up enough combined data to establish patterns that will further advance treatment. Drug trials could also be personalised by pinpointing which trial drug a patient is better suited to.
Over the next three years, more than 1,000 Victorians with rare and challenging cancers are set to benefit from genomic testing through an initiative led by Professor Grimmond.
“The Cancer of Unmet Need Initiative is piloting real-time testing of our most challenging cancer cases, integrating patients’ genomic data into routine clinical decision-making for a truly personalised approach to treatment and care,” Professor Grimmond says.
The $A6 million initiative is the first flagship project of a partnership announced in 2019 by University of Melbourne and Illumina, one of the world’s leading biotech companies.

Health & Medicine
Big data puts genomic handbrake on cancer
“While the overall outlook for people with cancer has almost doubled in recent decades, this initiative is targets those classes of cancers that have not seen these improved outcomes.
“It’s providing these patients with rapid diagnositics and a gateway to better cancer care and targeted clinical trials.”
Professor Grimmond and his team are already sequencing patients as part of the Victorian Comprehensive Cancer Centre’s (VCCC) Precision Oncology program.
The ultimate goal is for precision oncology to turn cancer into a manageable chronic disease, as well as minimising treatment failure and side effects.
For example, if a drug has an 80 per cent success rate but sequencing suggested you were one of the 20 per cent of patients that it would not work on, your doctor could direct you to another more suitable treatment.
“We want to take the guesswork out of therapy,” Professor Grimmond says.
“Ultimately, we want to help those at the forgotten fringes of cancer care on an individual level, to improve equity of patient outcomes.”
Banner: Getty Images