
Health & Medicine
Is this the end for alcohol handwash in hospitals?
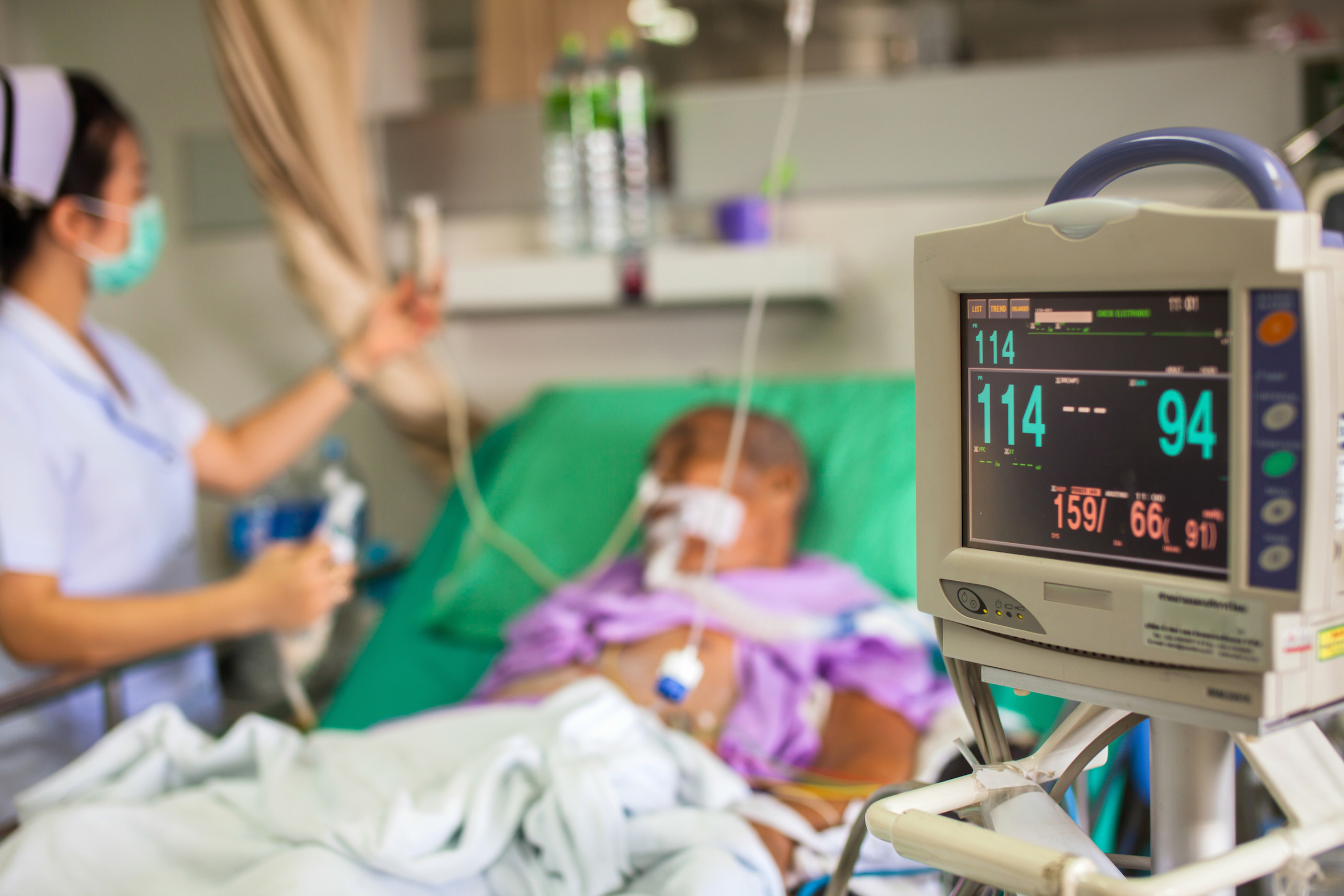
In parts of Europe, some strains of the latest superbug are already resistant to all known antibiotics – but researchers don’t yet know how it is spreading internationally
Published 4 September 2018
When Dr Jean Lee came across a case of an otherwise healthy patient who had become seriously ill from a hospital-acquired infection in 2012, she knew she had to investigate further.
Dr Lee, who was working as an infectious diseases registrar at a Melbourne hospital, became concerned when the patient experienced complications following a relatively simple elective procedure.

“He should have been in and out of hospital quickly,” she says. “But instead he contracted a very resistant infection and had to stay in hospital for over two months, followed by rehab.”
It turned out he had contracted Staphylococcus epidermidis (S. epidermidis), a bacteria found on human skin, and one of the most common causes of hospital associated infections.
It hasn’t previously been considered a major concern, with doctors more worried about other antibiotic-resistant superbugs like its cousin methicillin-resistant Staphylococcus aureus (MRSA) – commonly known as golden staph.

Health & Medicine
Is this the end for alcohol handwash in hospitals?
But, as Dr Lee discovered, S. epidermidis is becoming resistant to most antibiotics and – perhaps most worrying of all – in parts of Europe some strains are already resistant to all known antibiotics.
“Other bugs are more deadly, and this is a bug we all have on our skin, so I think there’s a perception it’s not quite so bad and that when a patient contracts it we can deal with it,” explains Dr Lee.
“But we’ve discovered resistance has got to the point where some of these cases are almost untreatable.”
After discovering her patient’s infection was caused by S. epidermidis, Dr Lee started a six-year project with her consultant and PhD supervisor, Professor Ben Howden, to understand how widespread its resistance is. Their findings have now been published in Nature Microbiology.
They identified three strains of the bacteria, which have become resistant to the two separate antibiotics currently used to treat infections – rifampicin and vancomycin.
Professor Howden, who remains a clinician and is also the Director of the Microbiological Diagnostic Unit Public Health Laboratory at the Doherty Institute, says this resistance is concerning because vancomycin in particular is used as a last line of defence against infections.
“Current guidelines for these types of infections recommend a combination treatment of rifampicin and vancomycin, which are completely unrelated and should protect against one another,” explains Professor Howden.
“But this finding reveals resistance to one antibiotic is causing resistance in the other, raising concerns about current treatment guidelines.
Health & Medicine
The chemical warfare against bacterial superbugs
“It has also managed to spread despite the good infection control now in place in hospitals in developed countries, which has helped manage golden staph.”
Dr Lee and Professor Howden first established that the resistant strains were present in hospitals in Victoria, and then expanded their search to determine where else they could be found.
They looked at hundreds of S. epidermidis cases from 78 institutions in ten countries around the world, and found they have spread globally.
“We worked with reference centres in the UK, Europe and America,” says Dr Lee. “These centres are sent difficult to treat or unusual bugs from individual hospital laboratories. We discovered these three new strains in difficult-to-treat cases in completely unrelated people.
“When we saw the infections exhibiting the same pattern of antibiotic resistance we realised the new resistant strains are everywhere.”
The researchers used the whole genome sequencing technology available at the Doherty Institute to rapidly sequence the bacteria’s whole genetic code.
“This meant we could compare our international samples at a genetic level to see how they were related,” explains Professor Howden. “We found the same mutations making bugs very antibiotic resistant in every country we tested.”
Patients tend to contract an S. epidermidis infection when they have a foreign object, like a line to administer drug or a catheter, inserted. Because the bug isn’t particularly virulent, infected patients also tend to be immunocompromised.
“When you break the skin you provide an entry portal for the bug, so an infection usually requires something that doesn’t belong in the body,” explains Dr Lee. “The bug creates a protective layer around whatever has been inserted, which allows it to avoid antibiotics and the body’s immune response.”
It is commonly found among patients in intensive care, because they tend to have so many lines inserted, and it is likely this has contributed to the spread of the resistant strains, says Professor Howden.
Health & Medicine
Enlisting nanoparticles in the fight against superbugs
“Catheters and other implanted devices are frequently impregnated with antibiotics as a strategy to prevent infection, however this approach may be promoting the development of resistance,” says Professor Howden.
“Also, these infections are most prevalent in intensive care, where patients are sickest and strong antibiotics are liberally prescribed, promoting the development of additional resistance.”
While the researchers say it’s likely these new strains of S. epidermidis are only acquired in hospital environments, they don’t yet know how it is spreading internationally.
“Like so many areas of science, this discovery has raised lots of new questions,” says Dr Lee.
“One of the most pressing is to work out exactly how the bug is being passed to patients. Knowing the mechanism will help us work out how it is spreading internationally.
“We also need to understand all the clinical outcomes – how much is the antibiotic resistance contributing to poor outcomes and what can be done about it.”
One of the first tasks is to revise treatment guidelines.
“We know what we’re currently doing is just leading to more resistance,” says Professor Howden. “So, we urgently need to think about what we should be recommending instead.
“There is an urgent need for an international monitoring system to understand the prevalence and impact of S. epidermidis and to systematically measure antibiotic resistance and infections due to this pathogen.”
Banner image: Shutterstock