Health & Medicine
Understanding epilepsy
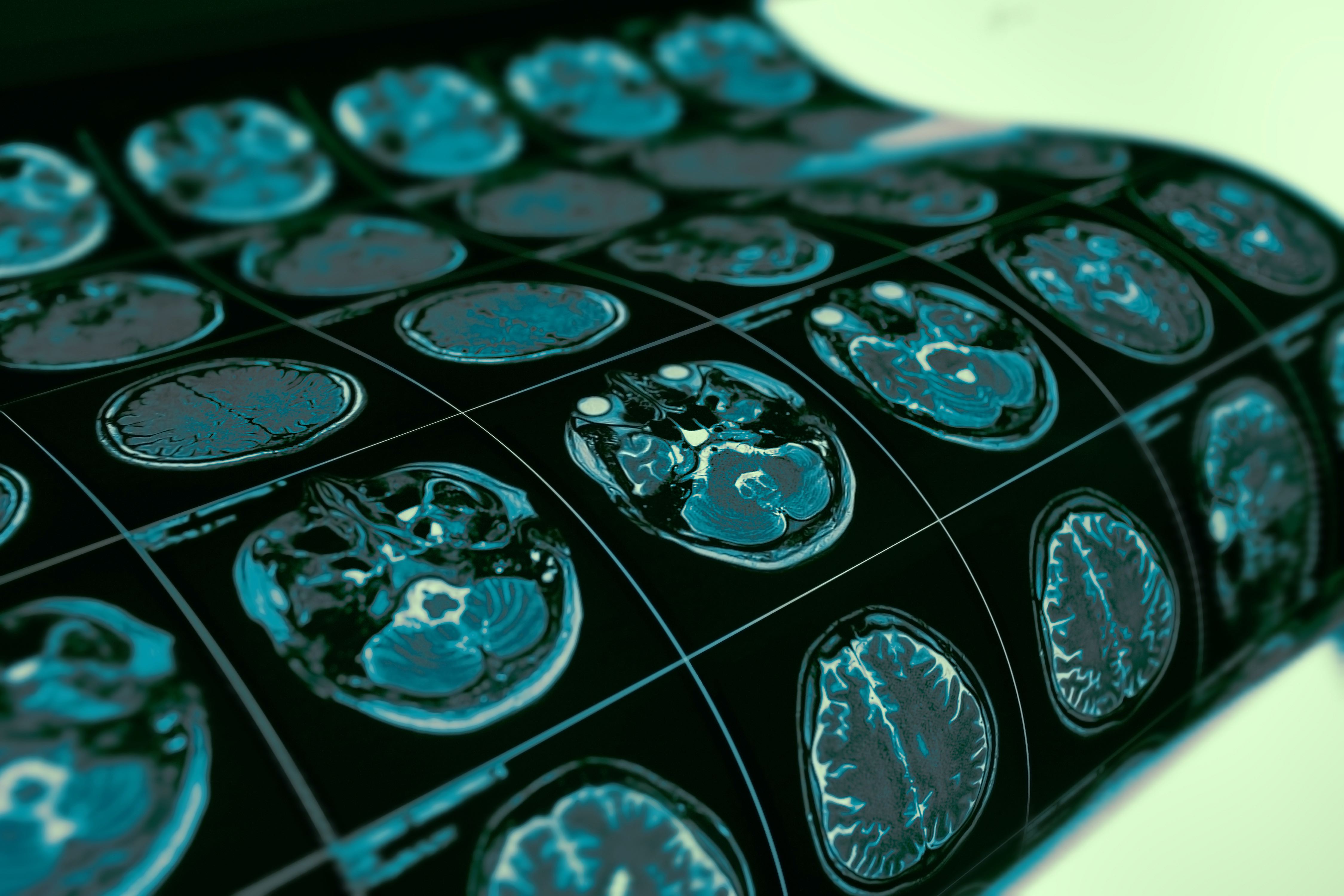
Associate Professor Reid is developing new drugs for epilepsy, focussing on both genetics and small molecules, to treat more people with epilepsy
Published 16 September 2020
Associate Professor Chris Reid was working as a hospital pharmacist when he saw a series of patients in a neurological ward who were not treatable.
“I thought well I can only do so much as a pharmacist. I would like to actually do something at a more fundamental level,” says Associate Professor Reid, Principal Research Fellow, member of Faculty and Head of the Neurophysiology of Excitable Networks Laboratory at the Florey Institute of Neuroscience and Mental Health.

Health & Medicine
Understanding epilepsy
“I’ve been very fortunate to be part of the genetic revolution which was started by Professor Sam Berkovic and Professor Ingrid Scheffer from the University of Melbourne. I joined Professor Steve Petrou’s lab at a time when that was very new.
“Things have moved incredibly quickly over the last 25 years, which is when the first epilepsy gene was discovered, to a point now where gene therapy is becoming a reality.”
Associate Professor Reid is currently developing a new treatment for epilepsy.
His research project is part of BioCurate, an independently-operated venture catalyst jointly formed by the University of Melbourne and Monash University.
“We do need alternative approaches as well – which is really the program that I’ve been driving with BioCurate. What we’ve done is actually identified a channel in the brain that’s in a hotspot that causes seizure generalisation. That’s when seizures cause the big tonic-clonic seizure that we mostly associate with epilepsy,” says Associate Professor Reid.
“We have a small molecule program to design drugs to that target with the view of treating a larger population of epilepsy people. So it’s a two-pronged approach, both of which have their risks, but both of which are moving forward particularly well.”
Episode recorded: August 6, 2020.
Interviewer: Dr Andi Horvath.
Producer, audio engineer and editor: Chris Hatzis.
Co-production: Silvi Vann-Wall and Dr Andi Horvath.
Banner image: Shutterstock