
Protecting the world from the threat of pandemics

Creating mathematical and computational models of infectious diseases like pandemic flu gives government and policy-makers a toolkit to respond to this ever-present threat
Published 14 March 2019
Bill Gates recently issued a stark warning to the world.
“We can’t predict when,” he told the Massachusetts Medical Society last year, “but...there is a significant probability of a large and lethal, modern-day pandemic occurring in our lifetimes.”
He has similarly reflected that while people rightly worry about dangers like terrorism and climate change “if anything is going to kill tens of millions of people in a short time, it will probably be a global epidemic. And the disease would most likely be a form of the flu, because flu spreads easily through the air.”

Indeed, Professor Jodie McVernon, the Director of Epidemiology at the Doherty Institute, notes that infectious diseases are an ever-present threat.
“With the increase of global travel and trade, new infections can spread rapidly across countries and continents,” she says.
And if you thought bird flu was no longer a threat, think again.

“Avian influenza remains one of the highest risks to human health,” she says, “with the possibility that new strains of flu could cross over from birds to humans and become infectious enough to cause a global pandemic.”
Professor McVernon is leading a research team that is collaborating with public health professionals to create mathematical and computational models of infectious diseases such as pandemic flu. Their research is directly informing public health policy on a global scale.
“My work aims to help public health decision-makers and policy-makers plan for these future unknowns,” she says.
“We develop models that simulate multiple possible pandemic scenarios of different infectiousness and severity and consider what their likely impacts will be on the health system and society.”
The models chart what might happen in an epidemic under a range of different scenarios.
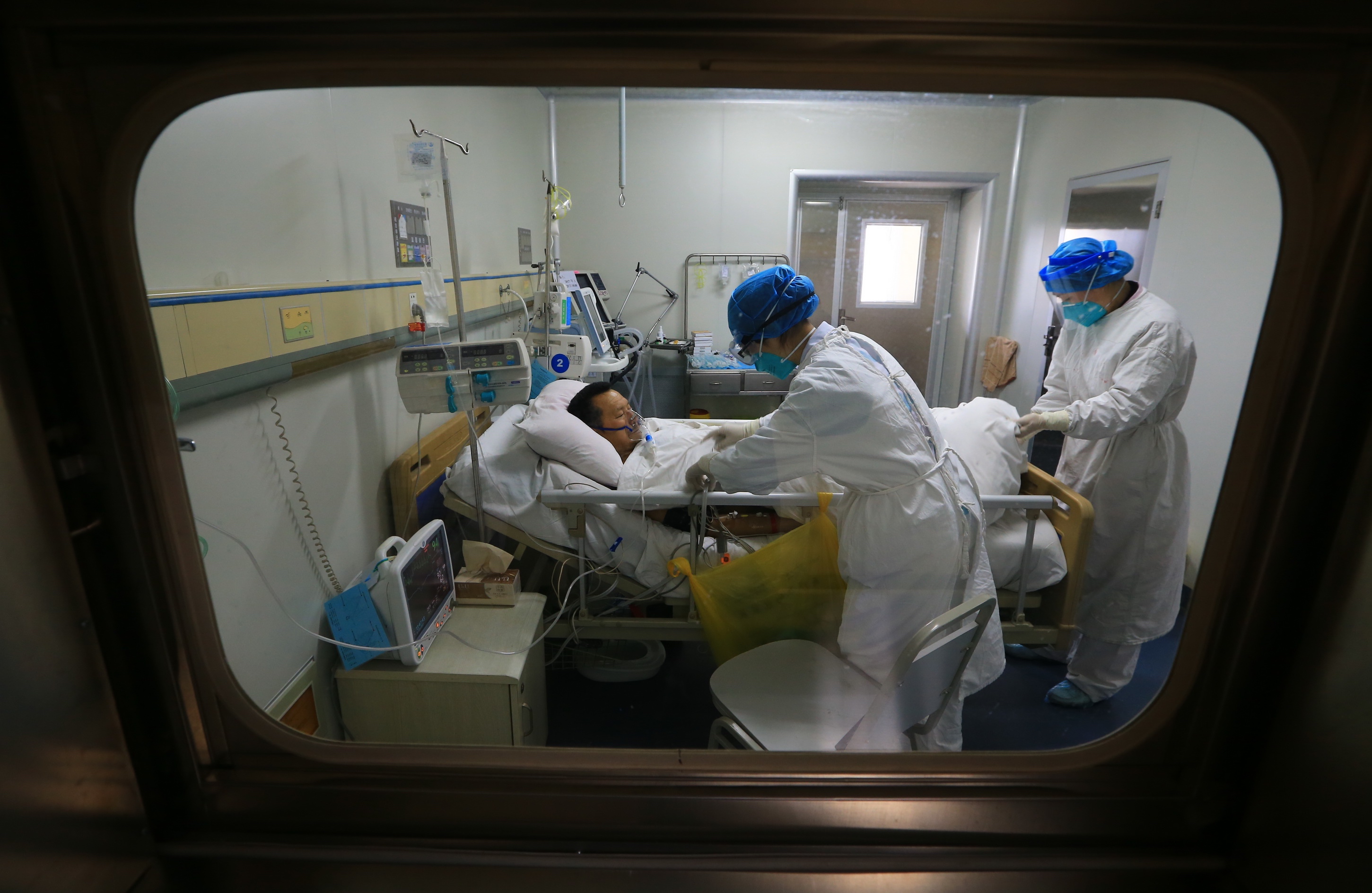
“I use a chunky Lego block analogy,” Professor McVernon explains. “We basically say people are either susceptible, infectious or recovered. We can then build up these simple Lego blocks into quite complex representations of society, to simulate an epidemic.”
They also help to guide thinking about the best strategies to use and inform decisions about stockpiling critical medicines and protective equipment.
“We model diseases like pandemic flu based on what we’ve observed in the past, to consider what we might see in the future. We then run many thousands of simulations and build in different measures to control disease like vaccinations and antiviral drugs, to see how they can best be used to prevent disease.”
Health & Medicine
Coming to grips with a ‘silent killer’
Thanks to the team’s research, Australia now has a necessary policy toolkit to prepare for and mitigate the whole-of-society risks posed by emergent infectious diseases. Professor McVernon’s research has helped inform the Australian Health Management Plan for Pandemic Influenza, which is one of the primary policy instruments with which the Australian government responds to emergent infectious disease threats.
“Collaboration is everything. With this approach, we’re helping to recommend the level of intervention necessary to protect health.”
The research team is taking that framing and thinking about capacity to deliver healthcare in Australia. How many hospital beds do we have, how many vaccines can we give out, and thinking through questions like: how many anti-viral drugs do we need in the stockpile? How do we use and distribute it?
The rapid spread of avian influenza, and recent outbreaks of the Ebola and Zika viruses, continue to threaten health across the world – particularly if new strains cross over to humans and lead to a global pandemic.

The need to prepare pandemic response plans that are both flexible and scalable is an urgent challenge for policy-makers and practitioners.
“The global community needs to be prepared for these threats, to be able to respond appropriately, to prevent sickness and death.”
The 1918 influenza pandemic, generally referred to as the greatest pandemic in history, killed as many as 100 million people, or 5 per cent of the global population. Today, Mr Gates has warned, “a flu as contagious and lethal as the 1918 one would kill nearly 33 million people in just six months.”
With the world now far more connected, the risk of a global pandemic has increased. But that very connection is also enabling researchers to collaborate globally in the search for big solutions to such a complex problem.
“Our work in Australia is part of a world-wide effort to be better prepared for infectious diseases. Infectious disease isn’t bound by borders, so the more aligned we are in thinking about information and methods, the better prepared we will be as a global community.”
Part of the problem has been that previous protection plans had been written for catastrophic outbreaks. Professor McVernon notes that when swine flu hit Australia in 2009, although it was serious, it ended up being “a lot milder than everybody expected. And there was nothing in the plan to help decision-makers know how to adjust their plan.”
Her team has helped create a Pandemic Impact Assessment Framework, which guides decision-makers to consider how infectious the disease is, and how rapidly it’s spreading.
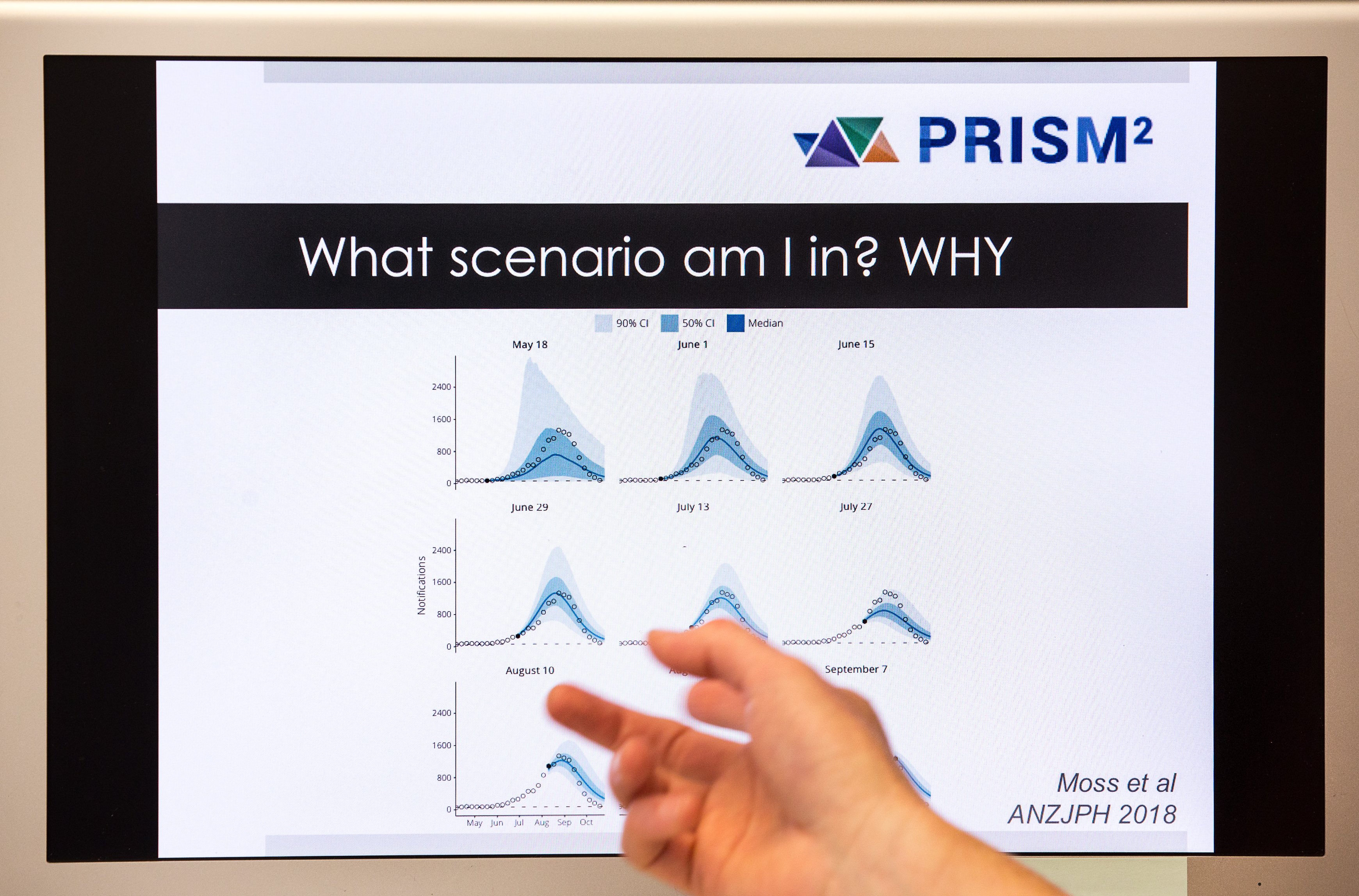
“These two things really determine how big and bad it is going to get,” she explains.
“Depending on that, you might go in all guns blazing, stopping the AFL Grand Final and shutting down schools, but if everyone’s not going to die then that would be completely excessive, and more disruptive to society than just telling people to stay at home if they feel sick, or maybe see their GP.”
The new framework has been developed alongside similar frameworks in other countries around the world, underscoring the need for global collaboration.
Professor McVernon compares her job to that of a translator, working closely with public health physicians and policy advisors across disciplines and sectors.

Health & Medicine
The flu-hunters
“I’m the person in the middle. My work relies on being able to synthesise a whole range of evidence that determines how an infection spreads in the population, from basic virology and immunology, through to sociology and human geography.”
She also advises the Australian government on developing ethical frameworks that help determine how best to use resources that might be in short supply at the beginning of an outbreak. The framework helps “determine the overall objectives of the response, including fairness and best outcomes for all,” she says.
“You might, for example, intuitively think we should give vaccines to everybody at risk. But the model might say it’s actually more effective, and quicker, to give it to school children, who spread it easily, and maybe you can prevent vulnerable people from getting sick in the first place.”
The long-term aim of the research is to combine disease surveillance and forecasting methods, so they can be built into routine public health decision support and response systems, and applied rapidly and effectively to real-world pandemics.
Banner: Supplied
