
Sciences & Technology
Close to the bone: The case for remodelling racehorse training
Dr Babatunde Ayodele is part of a team researching the bones of racehorses. Now, his research has inspired the façade of a new University building.
Published 5 April 2019
I had every intention of studying human medicine. I started my veterinary degree at the University of Ibadan in Nigeria and thought I would change over to human medicine in my second year. But there were opportunities to work in research and public health as a veterinarian. I enjoyed that, so I stayed.
After finishing my degree, I did national service. I was part of the National Youth Service Corps program in Nigeria where all graduates serve the government for one year. I was posted to Okitipupa in Ondo State, a very small town in an agricultural area.

My day normally started with a meat inspection at the abattoir and then I’d attend to animal patients, doing disease surveillance and control. There were no major outbreaks, but we reported a few tuberculosis cases to government.
I became interested in developmental conditions affecting bones. I was mostly influenced by the research that my supervisor at the time was doing on bones. My first experience of academic research was during my veterinary degree and during my national service, I then looked for further research opportunities and I saw that the University of Melbourne had a high ranking in the world.
I contacted Professor Eleanor Mackie at the University who was working on osteochondrosis – a developmental orthopaedic disease affecting rapidly growing animals, like horses, chickens, dogs and humans. Fortunately, I received a Graduate Research Scholarship and began my research career in Melbourne.

Sciences & Technology
Close to the bone: The case for remodelling racehorse training
Cartilage is made up of cells called chondrocytes which undergo different stages of development. And Professor Mackie’s team was studying the failure of normal bone formation during cartilage growth. The last stage of cellular activity, before bone is formed, is called chondrocyte hypertrophy. The failure of this stage of cartilage growth is associated with the development of osteochondrosis.
I wanted to understand the molecular mechanisms guiding the process of chondrocyte hypertrophy, and how the failure of that process may lead to osteochondrosis.
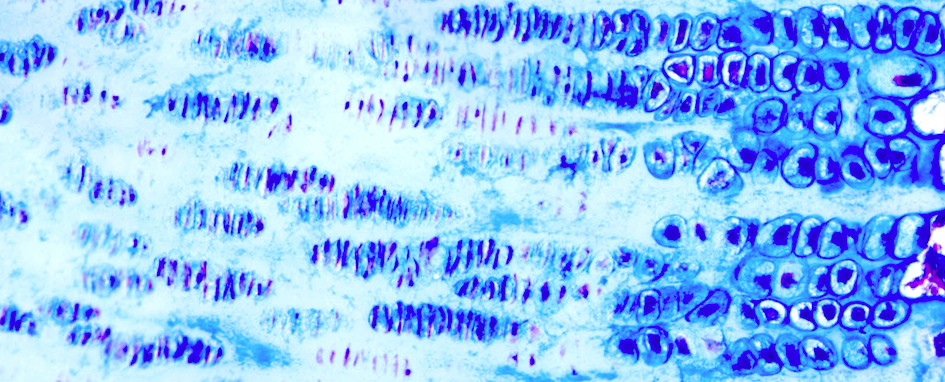
We use a lot of images to do our research. In order to see what’s happening we apply stains to tissue samples to enhance contrast. When we examine a slide under a microscope, we can more clearly see different components of the tissue and the arrangement of the cells that make up the tissue. The research identified a number of novel chondrocyte hypertrophy-associated genes.
When the University launched a competition for research images, specifically for bioscience academics, I had an image prepared. The idea was to use this image to influence the design of the façade of our new Western Edge Biosciences teaching building. I was so surprised when my image was selected.
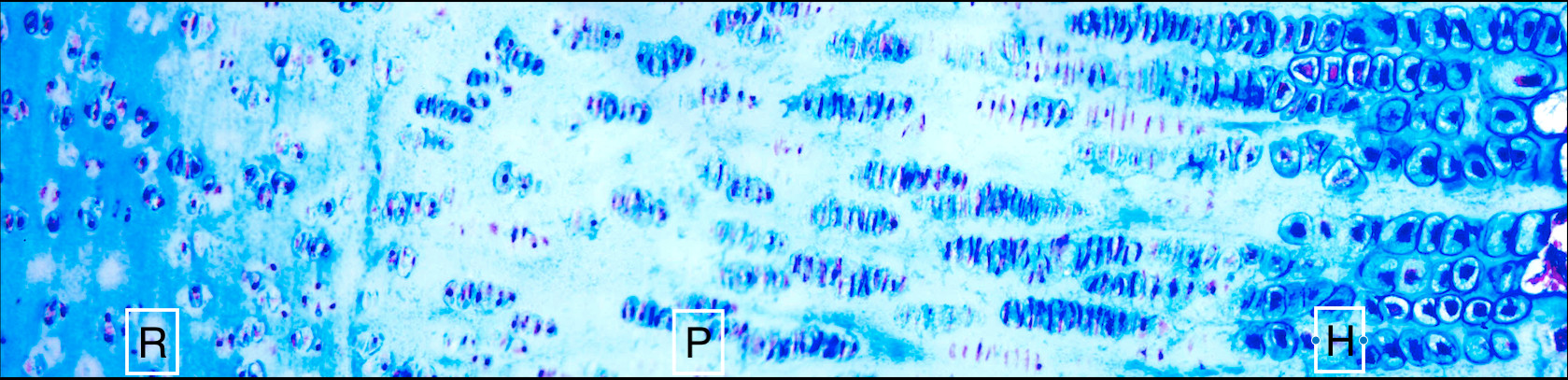
The image shows us a section of a horse growth cartilage, also called growth plate. It shows the chondrocytes – the cells that make up cartilage – at different stages of cellular activities. The area labelled ‘R’ shows the reserve or resting zone with fully differentiated chondrocytes. The area labelled ‘P’ shows proliferative chondrocytes, which are increasing in number and are arranged like stacks of coins.

The area marked ‘H’ shows the hypertrophic zone, where the chondrocytes become enlarged and space out before preparing the matrix to be replaced by bone. This process is called endochondral ossification and it drives our growth from when we’re a baby until adulthood. It’s the same process for many animals.
When I look at a growth cartilage, I see cellular activities and not the aesthetics. Incorporating the image into the design of the building is interesting because it’s impressive to see the design that emerges from this image and it makes me proud of what we as researchers do.
Now I’m part of a team looking at structural changes in the bones of racehorses. The Equine Limb Injury Prevention Program wants to understand how exercise and training affects the structure of bone and I’m working with veterinarians, epidemiologists, biomechanical engineers and other musculoskeletal biology researchers. We’re analysing sections of bone from horses at different stages of training and rest.

We want to develop a model to help trainers reduce the risk of bone and joint injuries in racehorses. Bone is a living, flexible tissue, it’s not static. Regions of bone sustain microfractures during exercise, which are resorbed and replaced during rest periods.
This process, known as remodelling, renews and maintains the strength of bone. But at the moment there’s no way of knowing the amount of microdamage that intensive exercise causes in bone because we can’t measure microdamage in living animals with current imaging techniques.

Sciences & Technology
Global food security under threat from crop and livestock diseases
Better understanding animals can help human beings. Cellular activities are similar, and what we understand of the molecular mechanisms regulating these activities in animals can be useful for humans as well.
I’d like to develop better ways to predict the risk of injury in equine athletes (like racehorses), possibly with the use of biomarkers. They are measurable elements in a biological system that indicate a pathological process or disease is occurring.
If we can better understand the mechanisms that lead to microdamage accumulation in bone and the development of bone fatigue, we’d be better at predicting the risk of injuries.
This understanding in horses may in turn be valuable for the bone health of other animal athletes and humans, too.
- As told to Stuart Winthrope