The genetic backburn

A counter-intuitive idea to introduce more of an invasive species in order to halt their spread. It might just work.
Published 24 February 2016
The best way to stop the spread of an invasive species might be to introduce more of them ahead of the invasion front. That makes absolutely no sense, right?
Well, we decided to test this idea using computer simulations and our study, published in the journal Proceedings of the Royal Society B, suggests it might not be as crazy as it first seems.
The key to this apparently nonsensical idea is that invasive species evolve; and fast. Studies from spreading populations around the world repeatedly show that invasion selects for increased dispersal ability.
The reason this happens is a phenomenon known as the Olympic Village effect. When high performing athletes spend time together, they are likely to breed together, and so some of their offspring are likely to be even better athletes than their parents.

Similarly, only the best invaders, the most dispersive individuals, will be on the invasion front each generation. Because these ‘high-performing athletes’ all find themselves at the same place at the same time, they breed with each other and their offspring tend to be more dispersive as a consequence.
So invasion fronts tend to move faster as time goes on. This explains, for example, why cane toads currently spread at around 50 km per year, whereas when they were first introduced, they spread at only one-fifth of that speed.
This tendency towards speed on the invasion front means that, as invasions progress, they not only accelerate, but they become harder to stop at natural landscape barriers.
A 30km wide strip of desert would have stopped the toad invasion in 1940, but not now.
Now the highly dispersive invasion front toads would charge across that barrier without a moment’s pause.
So what if we took some of the slow-dispersing individuals (from well behind the invasion front) and released them on the nearside of a landscape barrier?
Our simulations, using computer modeling, suggest that this ‘genetic backburn’, under the right circumstances, can make landscape barriers substantially more effective.
The less dispersive individuals that we introduce can’t breach the barrier and, through population growth and competition, they effectively exclude the highly dispersive individuals from ever reaching the barrier.
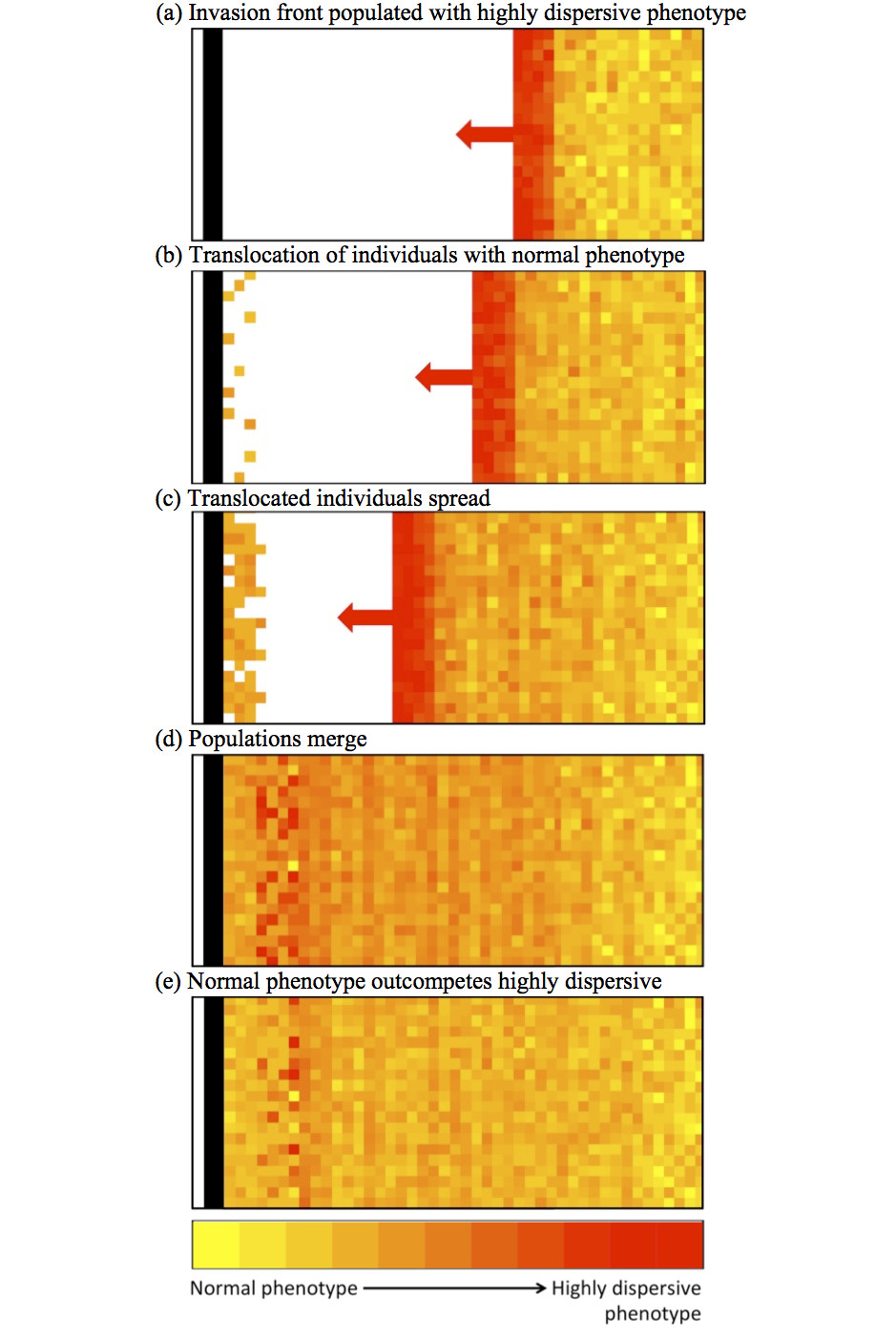
In the above diagram an invasion is progressing (from right to left) towards a landscape barrier (the black bar). The invasion front is made up of highly-dispersive individuals that can breach the barrier. Well behind the invasion front, however, are individuals that are substantially less dispersive.
We translocate some of these individuals to the nearside of our barrier, they start an invasion back towards the oncoming invasion front and ultimately prevent highly dispersive genotypes ever reaching the barrier.
So would managers ever undertake such a strategy? Well, that would probably depend on how well they understood and communicated the reasoning behind it.
Our analysis suggests that such an action never makes it more likely that a barrier will be breached, so genetic backburning can only help. The potential risk, however, is that if the barrier fails anyway then the invasive species will get to the far side of the barrier a little earlier than it otherwise would have.
How this risk is weighed against the benefit will be particular to each case.
In Australia, for example, we have the very real possibility of keeping cane toads out of the 270,000 km2 Pilbara region of Western Australia by using a dry landscape barrier – a strip of land with no permanent water sources.
Toads will probably reach the barrier in about a decade, and we have little doubt that the barrier would be greatly enhanced by use of a genetic backburn.
Given public sentiment towards toads, it is more or less inconceivable that we would be allowed to introduce them ten years ahead of the invasion front. But what about five years? Three?
These are difficult questions, but in the light of the advantages of genetic backburning they are questions we should ask sooner rather than later.
Banner Image: A cane toad at home on the grass. Picture: Flickr

