Health & Medicine
Your protective Igs: The major focus of COVID-19 vaccines

A Melbourne-based startup has built an mRNA platform that Australian scientists can use to accelerate their research
Published 25 February 2022
The story of messenger RNA (mRNA) begins with one of the classiest moves in scientific history.
In early 1961, three scientists —Sydney Brenner, François Jacob and Matt Meselson—submitted a paper to the journal Nature, detailing their groundbreaking discovery of a molecule they dubbed “messenger” RNA – or mRNA for short.

But they weren’t the only ones to have just discovered mRNA; Professor James Watson’s British/American research group – of Watson and Crick DNA double-helix fame – had too. He asked the trio to delay publication until his group had also submitted their own paper.
Scientific breakthroughs typically result in a frenetic race to publish first, but Professor Brenner and his colleagues generously agreed, and both papers appeared back-to-back in a May 1961 edition of Nature.
We have come a long way since then.

Health & Medicine
Your protective Igs: The major focus of COVID-19 vaccines
After decades of being dismissed as a scientific backwater, mRNA now represents arguably our best hope for combating the COVID-19 pandemic – thanks to the success of the Moderna and Pfizer/BioNTech vaccines in protecting many of us from the virus.
Much of mRNA’s appeal lies in its versatility and programmability, which has the potential to both compress regulatory timelines and drive down development costs.
And this reaches far beyond COVID-19 – and even beyond the realm of infectious diseases.
At its core, an mRNA molecule is simply a written instruction for a cell to manufacture a specific protein. And by stringing together each of four different chemical bases—adenine, guanine, cytosine and uracil—in different arrangements, it can be programmed to encode virtually any protein imaginable.

My research, in collaboration with other scientists, aims to apply this technology to the treatment of monogenic diseases, which are caused by harmful mutations within a single gene – for example, Niemann-Pick disease type C1 (NP-C1) is one of these.
Patients with NP-C1 have an impaired ability to process cholesterol within their cells as a result of an improperly functioning gateway protein, often leading to severe clinical symptoms affecting the brain, liver, spleen, and, in some cases, the lungs.
Our theory was that by supplying cells with mRNA coding for a functional version of this protein, we could potentially restore healthy protein levels, normalise cholesterol processing, and remedy the downstream effects of the disease.
Sure enough, our initial experiments using patient cells have borne out our hypothesis. We are now taking the next steps to develop a delivery system that can transport mRNA to disease-affected organs, like the brain.
Sciences & Technology
Live cell DNA architecture in real time
If successful, the same technology could be applied to a whole host of other monogenic diseases like Friedreich’s ataxia and Rett Syndrome — and there are thousands more of them.
mRNA is fast becoming a molecule of interest to scientists across the biomedical research community.
But it goes further.
Australia’s state and federal governments have negotiated the establishment of domestic mRNA manufacturing capacity here, but investment like this requires a constant, rich flow of mRNA candidates coming into the pipeline to ensure ongoing commercial feasibility.
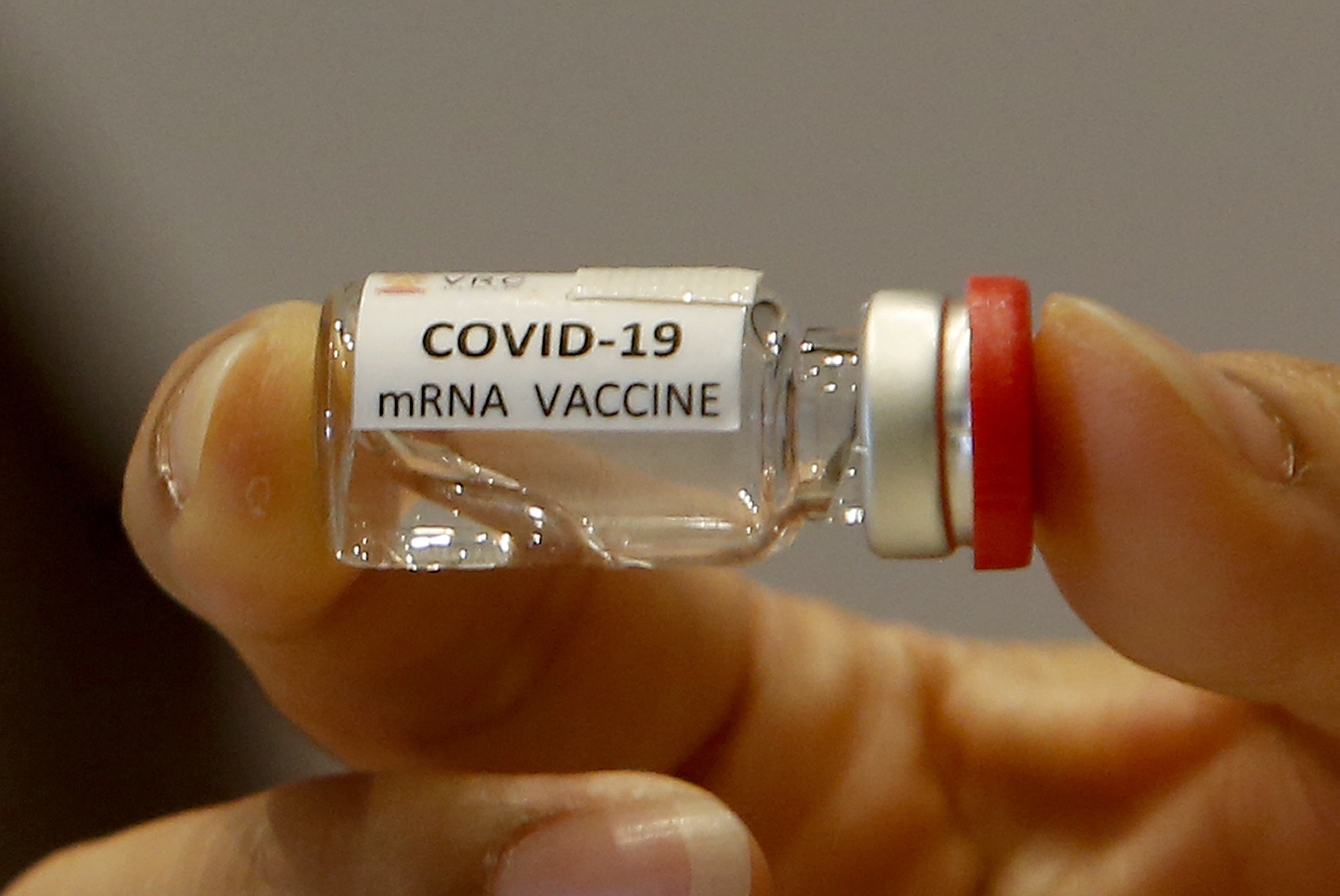
Realising that our research findings could be generalised to benefit others, I recently partnered with Professor Frank Caruso, head of the Caruso Nanoengineering Group at the University, to launch a new startup called Messenger Bio.
The core mission of the startup is to democratise access to frontier technologies, starting with mRNA, so that researchers anywhere in the world can get up to speed on new projects faster and more affordably than ever before.
We’re currently doing this by offering custom mRNA synthesis and lipid nanoparticle formulation.

Sciences & Technology
Who owns your DNA?
Previously, only highly specialised mRNA experts could work in this field. But by building out infrastructure that takes care of the end-to-end design, production, purification and formulation of mRNA, this technology can now be made available to anyone who wants to work with it.
Our longer-term vision is to build a platform that can serve scientists at all stages of their research journey – from bench to bedside.
Research like ours has just begun to scratch the surface of mRNA’s potential.
Soon, the technology could be used clinically in applications ranging from cancer immunotherapy and allergy research to protein replacement, genome and epigenome editing – and even stem cell reprogramming, which could see human cells instructed to repair damaged tissues in conditions like Parkinson’s disease.
The opportunity to shape the future of genetic medicine is well and truly upon us – and now is the time to be a part of the mRNA revolution.
Banner: Shutterstock